QUESTION IMAGE
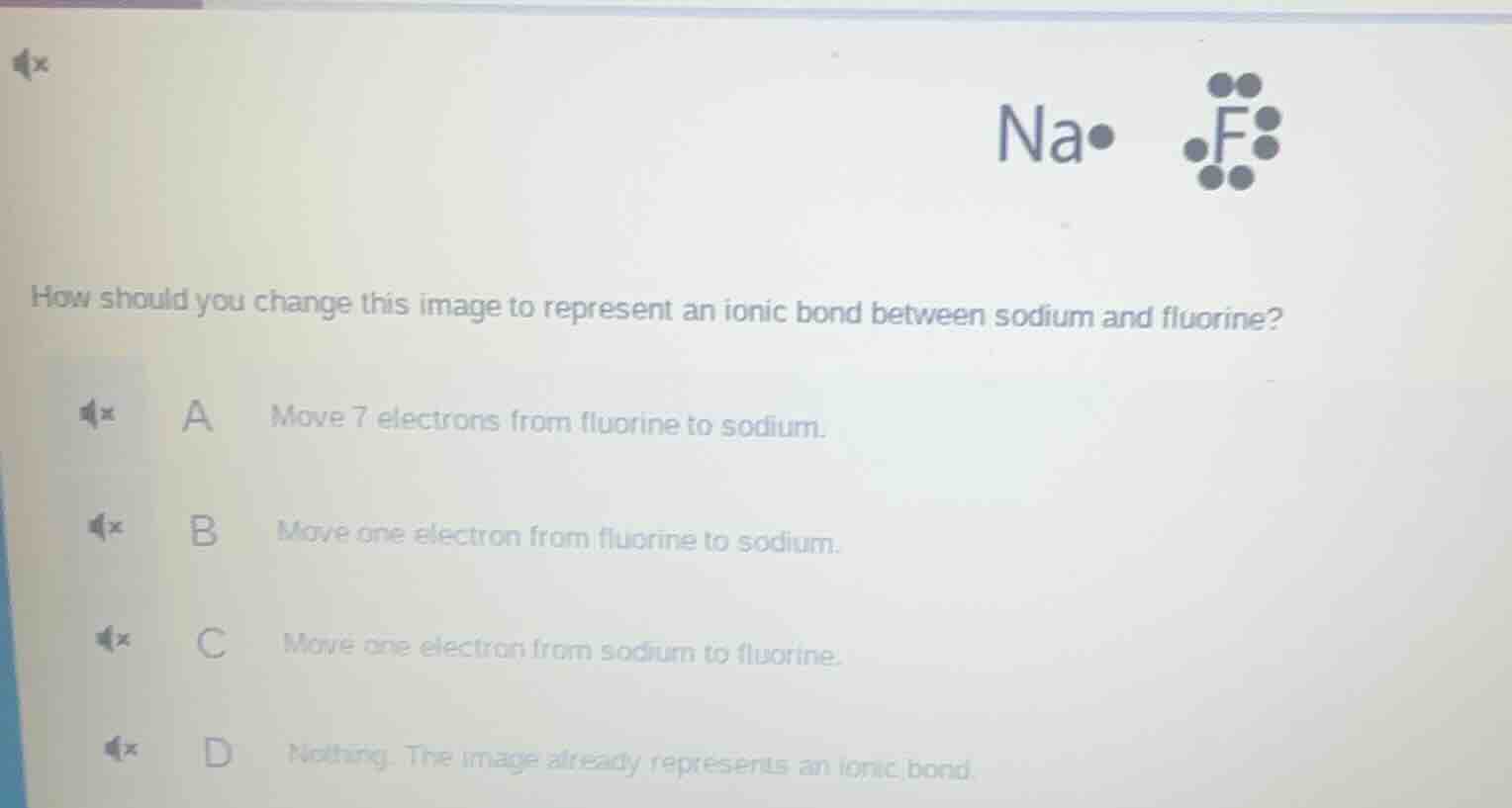
Question
how should you change this image to represent an ionic bond between sodium and fluorine? a move 7 electrons from fluorine to sodium. b move one electron from fluorine to sodium. c move one electron from sodium to fluorine. d nothing. the image already represents an ionic bond.
Ionic bonds form when a metal transfers valence electrons to a nonmetal. Sodium (Na) is a metal with 1 valence electron, and fluorine (F) is a nonmetal with 7 valence electrons. To achieve stable electron configurations, sodium donates its single valence electron to fluorine, forming positively charged Na⁺ and negatively charged F⁻, which attract to form an ionic bond.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Move one electron from sodium to fluorine.