QUESTION IMAGE
Question
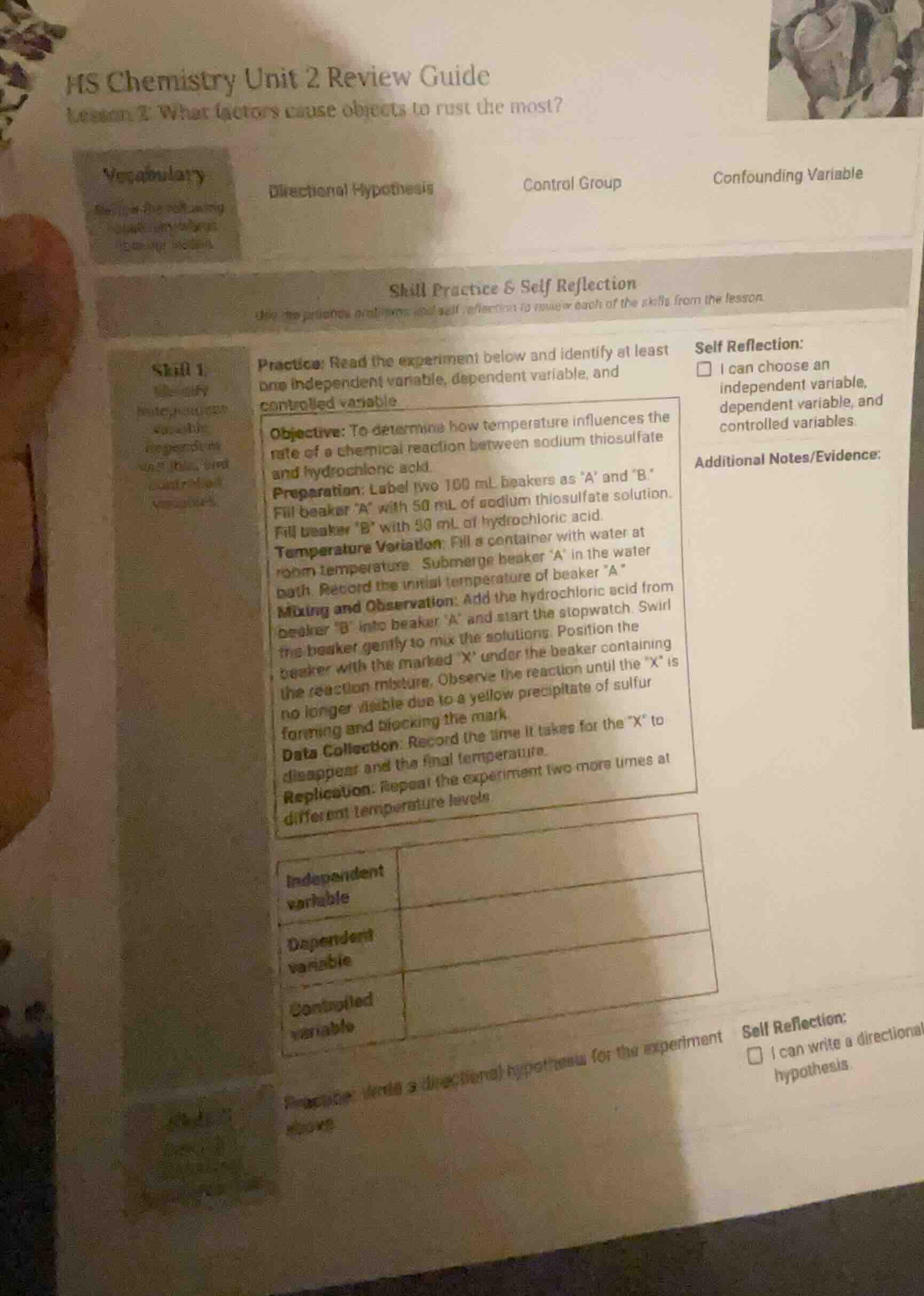
hs chemistry unit 2 review guide
lesson 2: what factors cause objects to rust the most?
vocabulary
directional hypothesis control group confounding variable
define the following vocabulary terms
skill practice & self reflection
use the practice problems and self - reflection to review each of the skills from the lesson.
skill 1:
identify independent, dependent, and controlled variables.
practice: read the experiment below and identify at least one independent variable, dependent variable, and controlled variable.
objective: to determine how temperature influences the rate of a chemical reaction between sodium thiosulfate and hydrochloric acid.
preparation: label two 100 ml beakers as “a” and “b.” fill beaker “a” with 50 ml of sodium thiosulfate solution. fill beaker “b” with 20 ml of hydrochloric acid.
temperature variation: fill a container with water at room temperature. submerge beaker “a” in the water bath. record the initial temperature of beaker “a.”
mixing and observation: add the hydrochloric acid from beaker “b” into beaker “a” and start the stopwatch. swirl the beaker gently to mix the solutions. position the beaker with the marked “x” under the beaker containing the reaction mixture. observe the reaction until the “x” is no longer visible due to a yellow precipitate of sulfur forming and blocking the mark.
data collection: record the time it takes for the “x” to disappear and the final temperature.
replication: repeat the experiment two more times at different temperature levels.
independent variable
dependent variable
controlled variable
practice: write a directional hypothesis for the experiment above
self reflection:
☐ i can choose an independent variable, dependent variable, and controlled variables.
additional notes/evidence:
☐ i can write a directional hypothesis.
Step 1: Identify the Independent Variable
The independent variable is the factor that is intentionally changed in an experiment. In this experiment, the objective is to see how temperature influences the reaction rate. So, the independent variable is the temperature of the reaction mixture (since it's the factor being varied across trials).
Step 2: Identify the Dependent Variable
The dependent variable is the factor that changes in response to the independent variable. Here, we are measuring the time it takes for the "X" to disappear (which is related to the reaction rate). So the dependent variable is the time taken for the "X" to disappear (or the reaction rate, as time is inversely related to rate).
Step 3: Identify Controlled Variables
Controlled variables are factors kept constant to ensure a fair test. From the experiment:
- The volume of sodium thiosulfate solution (50 mL) and hydrochloric acid (50 mL) are kept constant.
- The type of beakers (100 mL beakers) and the method of mixing (swirling gently) are also controlled. One example of a controlled variable is the volume of the reactant solutions (e.g., 50 mL of sodium thiosulfate and 50 mL of HCl).
Filling the Table:
| Variable Type | Value/Description |
|---|---|
| Dependent Variable | Time taken for the "X" to disappear (or the reaction rate of sodium thiosulfate and hydrochloric acid reaction) |
| Controlled Variable | Volume of reactant solutions (e.g., 50 mL of sodium thiosulfate, 50 mL of HCl), type of beakers (100 mL), mixing method, etc. (any one controlled variable can be listed, e.g., volume of sodium thiosulfate solution) |
For the Directional Hypothesis:
A directional hypothesis predicts the direction of the relationship between variables. Since the experiment tests how temperature affects reaction rate, and generally, increasing temperature increases reaction rate (faster reaction means less time for "X" to disappear):
Directional Hypothesis: As the temperature of the reaction mixture (containing sodium thiosulfate and hydrochloric acid) increases, the time taken for the "X" mark to disappear (reaction rate) will decrease (or the reaction rate will increase).
Final Answers for the Table (Example):
- Independent Variable: Temperature of the reaction mixture
- Dependent Variable: Time taken for the "X" to disappear
- Controlled Variable: Volume of sodium thiosulfate solution (50 mL) (or any other controlled variable from the list)
- Directional Hypothesis: If the temperature of the reaction between sodium thiosulfate and hydrochloric acid increases, then the time taken for the "X" to disappear will decrease because higher temperatures increase the kinetic energy of particles, leading to a faster reaction rate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step 1: Identify the Independent Variable
The independent variable is the factor that is intentionally changed in an experiment. In this experiment, the objective is to see how temperature influences the reaction rate. So, the independent variable is the temperature of the reaction mixture (since it's the factor being varied across trials).
Step 2: Identify the Dependent Variable
The dependent variable is the factor that changes in response to the independent variable. Here, we are measuring the time it takes for the "X" to disappear (which is related to the reaction rate). So the dependent variable is the time taken for the "X" to disappear (or the reaction rate, as time is inversely related to rate).
Step 3: Identify Controlled Variables
Controlled variables are factors kept constant to ensure a fair test. From the experiment:
- The volume of sodium thiosulfate solution (50 mL) and hydrochloric acid (50 mL) are kept constant.
- The type of beakers (100 mL beakers) and the method of mixing (swirling gently) are also controlled. One example of a controlled variable is the volume of the reactant solutions (e.g., 50 mL of sodium thiosulfate and 50 mL of HCl).
Filling the Table:
| Variable Type | Value/Description |
|---|---|
| Dependent Variable | Time taken for the "X" to disappear (or the reaction rate of sodium thiosulfate and hydrochloric acid reaction) |
| Controlled Variable | Volume of reactant solutions (e.g., 50 mL of sodium thiosulfate, 50 mL of HCl), type of beakers (100 mL), mixing method, etc. (any one controlled variable can be listed, e.g., volume of sodium thiosulfate solution) |
For the Directional Hypothesis:
A directional hypothesis predicts the direction of the relationship between variables. Since the experiment tests how temperature affects reaction rate, and generally, increasing temperature increases reaction rate (faster reaction means less time for "X" to disappear):
Directional Hypothesis: As the temperature of the reaction mixture (containing sodium thiosulfate and hydrochloric acid) increases, the time taken for the "X" mark to disappear (reaction rate) will decrease (or the reaction rate will increase).
Final Answers for the Table (Example):
- Independent Variable: Temperature of the reaction mixture
- Dependent Variable: Time taken for the "X" to disappear
- Controlled Variable: Volume of sodium thiosulfate solution (50 mL) (or any other controlled variable from the list)
- Directional Hypothesis: If the temperature of the reaction between sodium thiosulfate and hydrochloric acid increases, then the time taken for the "X" to disappear will decrease because higher temperatures increase the kinetic energy of particles, leading to a faster reaction rate.