QUESTION IMAGE
Question
Question was provided via image upload.
Since the problem is not clearly stated (e.g., identifying the compounds, their functional groups, or another task), I'll assume a common task like naming the compounds or identifying functional groups. Let's analyze each structure:
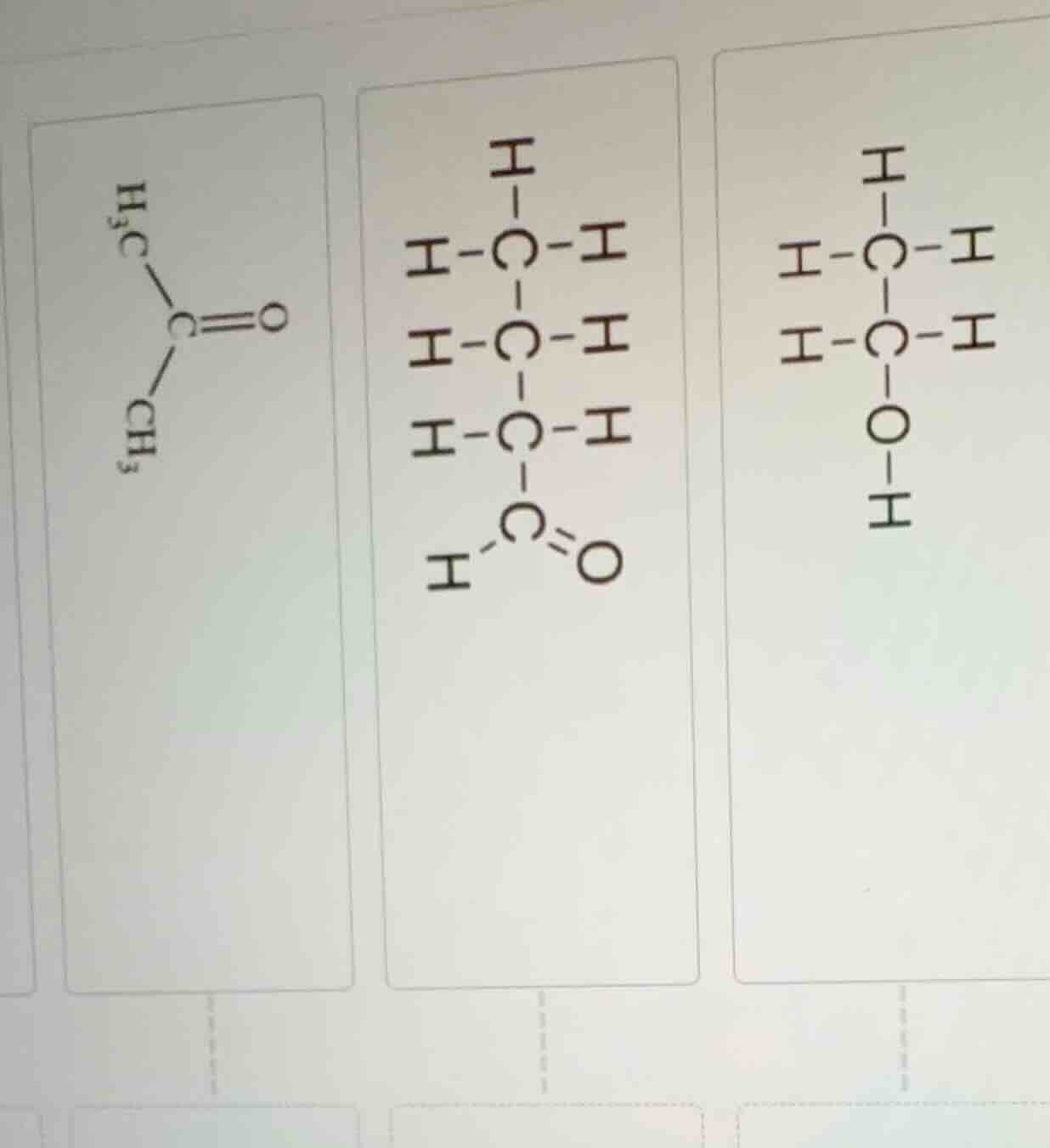
First Structure (Left):
- Formula: \( \text{CH}_3\text{COCH}_3 \) (Acetone, Propanone)
- Functional Group: Ketone (\( \text{C}=\text{O} \) bonded to two alkyl groups)
Second Structure (Middle):
- Formula: \( \text{CH}_3\text{CH}_2\text{CH}_2\text{CHO} \) (Butanal)
- Functional Group: Aldehyde (\( \text{C}=\text{O} \) bonded to a hydrogen and an alkyl group)
Third Structure (Right):
- Formula: \( \text{C}_2\text{H}_5\text{OH} \) (Ethanol)
- Functional Group: Alcohol (\( -\text{OH} \) group bonded to a carbon)
If the task was to identify the functional groups:
- Left: Ketone
- Middle: Aldehyde
- Right: Alcohol
If you meant another task (like naming, molecular formula calculation), please clarify. For molecular formula calculation (e.g., third structure: \( \text{C}_2\text{H}_6\text{O} \)):
Step 1: Count atoms
- Carbon (\( \text{C} \)): 2 (from two \( \text{C} \) atoms)
- Hydrogen (\( \text{H} \)): 6 (each \( \text{C} \) has 3, but one \( \text{C} \) has \( -\text{OH} \), so \( 3 + 3 = 6 \))
- Oxygen (\( \text{O} \)): 1 (from \( -\text{OH} \))
Step 2: Write formula
\( \text{C}_2\text{H}_6\text{O} \)
Since the problem is unclear, please provide more details (e.g., "Identify the functional group of the right structure" or "Name the left compound") to get a precise answer.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Since the problem is not clearly stated (e.g., identifying the compounds, their functional groups, or another task), I'll assume a common task like naming the compounds or identifying functional groups. Let's analyze each structure:
First Structure (Left):
- Formula: \( \text{CH}_3\text{COCH}_3 \) (Acetone, Propanone)
- Functional Group: Ketone (\( \text{C}=\text{O} \) bonded to two alkyl groups)
Second Structure (Middle):
- Formula: \( \text{CH}_3\text{CH}_2\text{CH}_2\text{CHO} \) (Butanal)
- Functional Group: Aldehyde (\( \text{C}=\text{O} \) bonded to a hydrogen and an alkyl group)
Third Structure (Right):
- Formula: \( \text{C}_2\text{H}_5\text{OH} \) (Ethanol)
- Functional Group: Alcohol (\( -\text{OH} \) group bonded to a carbon)
If the task was to identify the functional groups:
- Left: Ketone
- Middle: Aldehyde
- Right: Alcohol
If you meant another task (like naming, molecular formula calculation), please clarify. For molecular formula calculation (e.g., third structure: \( \text{C}_2\text{H}_6\text{O} \)):
Step 1: Count atoms
- Carbon (\( \text{C} \)): 2 (from two \( \text{C} \) atoms)
- Hydrogen (\( \text{H} \)): 6 (each \( \text{C} \) has 3, but one \( \text{C} \) has \( -\text{OH} \), so \( 3 + 3 = 6 \))
- Oxygen (\( \text{O} \)): 1 (from \( -\text{OH} \))
Step 2: Write formula
\( \text{C}_2\text{H}_6\text{O} \)
Since the problem is unclear, please provide more details (e.g., "Identify the functional group of the right structure" or "Name the left compound") to get a precise answer.