QUESTION IMAGE
Question
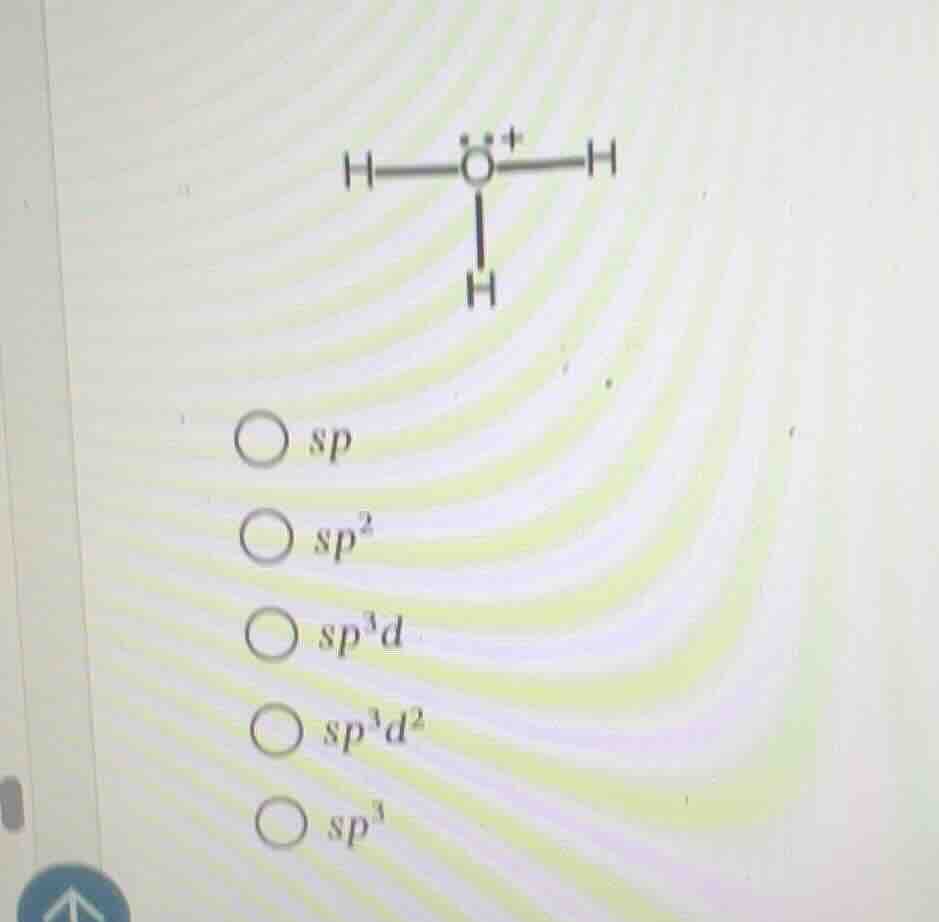
the image shows a chemical structure of a molecule with a central o atom bonded to three h atoms (with a positive charge on o) and a lone pair of electrons. below the structure, there are multiple - choice options for hybridization: sp, sp², sp³d, sp³d², sp³.
Step1: Count electron domains
The central oxygen atom has 3 bonding pairs (to 3 H atoms) and 1 lone pair, so total electron domains = $3 + 1 = 4$.
Step2: Match to hybridization
4 electron domains correspond to $sp^3$ hybridization.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{sp^3}$