QUESTION IMAGE
Question
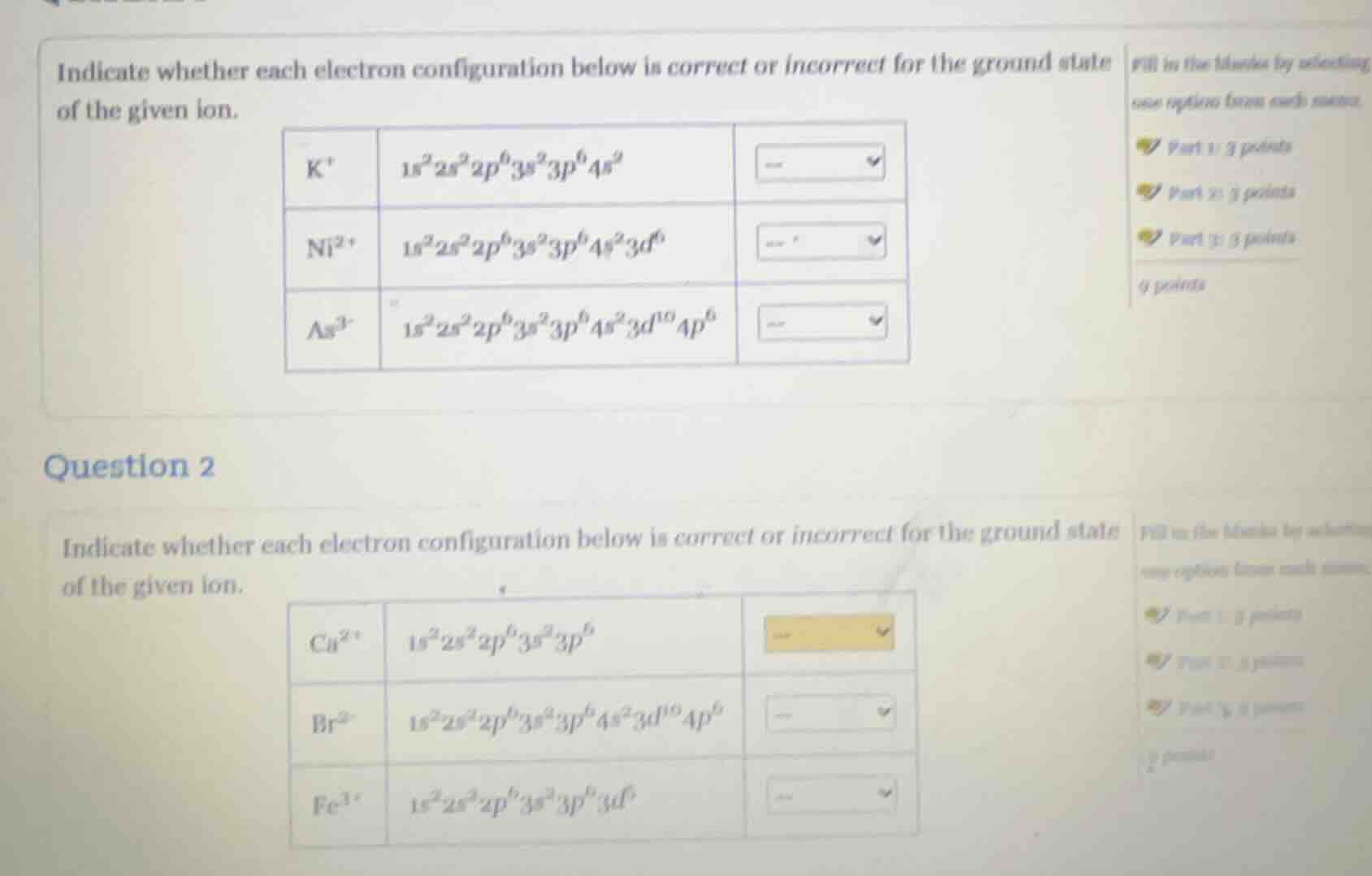
indicate whether each electron configuration below is correct or incorrect for the ground state of the given ion.
k⁺: 1s²2s²2p⁶3s²3p⁶4s²
ni²⁺: 1s²2s²2p⁶3s²3p⁶4s²3d⁶
as³⁻: 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶
question 2
indicate whether each electron configuration below is correct or incorrect for the ground state of the given ion.
cu²⁺: 1s²2s²2p⁶3s²3p⁶
br²⁻: 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁶
fe³⁺: 1s²2s²2p⁶3s²3p⁶3d⁵
Step1: Analyze $\text{K}^+$ electron config
Neutral K: $1s^22s^22p^63s^23p^64s^1$. $\text{K}^+$ loses 1 $4s$ electron: $1s^22s^22p^63s^23p^6$. Given config is $1s^22s^22p^63s^23p^64s^2$, so incorrect.
Step2: Analyze $\text{Ni}^{2+}$ electron config
Neutral Ni: $1s^22s^22p^63s^23p^64s^23d^8$. $\text{Ni}^{2+}$ loses 2 $4s$ electrons: $1s^22s^22p^63s^23p^63d^8$. Given config is $1s^22s^22p^63s^23p^64s^23d^6$, so incorrect.
Step3: Analyze $\text{As}^{3-}$ electron config
Neutral As: $1s^22s^22p^63s^23p^64s^23d^{10}4p^3$. $\text{As}^{3-}$ gains 3 $4p$ electrons: $1s^22s^22p^63s^23p^64s^23d^{10}4p^6$. Given config matches, so correct.
Step4: Analyze $\text{Cu}^{2+}$ electron config
Neutral Cu: $1s^22s^22p^63s^23p^64s^13d^{10}$. $\text{Cu}^{2+}$ loses 1 $4s$ and 1 $3d$ electron: $1s^22s^22p^63s^23p^63d^9$. Given config is $1s^22s^22p^63s^23p^6$, so incorrect.
Step5: Analyze $\text{Br}^{2-}$ electron config
Neutral Br: $1s^22s^22p^63s^23p^64s^23d^{10}4p^5$. $\text{Br}^{2-}$ would gain 2 electrons, but Br only gains 1 to reach noble gas config; $4p$ can hold max 6, so $1s^22s^22p^63s^23p^64s^23d^{10}4p^7$ is invalid. Given config is $1s^22s^22p^63s^23p^64s^23d^{10}4p^6$, which is $\text{Br}^-$, so incorrect for $\text{Br}^{2-}$.
Step6: Analyze $\text{Fe}^{3+}$ electron config
Neutral Fe: $1s^22s^22p^63s^23p^64s^23d^6$. $\text{Fe}^{3+}$ loses 2 $4s$ and 1 $3d$ electron: $1s^22s^22p^63s^23p^63d^5$. Given config matches, so correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{K}^+$: Incorrect
- $\text{Ni}^{2+}$: Incorrect
- $\text{As}^{3-}$: Correct
- $\text{Cu}^{2+}$: Incorrect
- $\text{Br}^{2-}$: Incorrect
- $\text{Fe}^{3+}$: Correct