QUESTION IMAGE
Question
ionic bonding 6.1-6.3 test
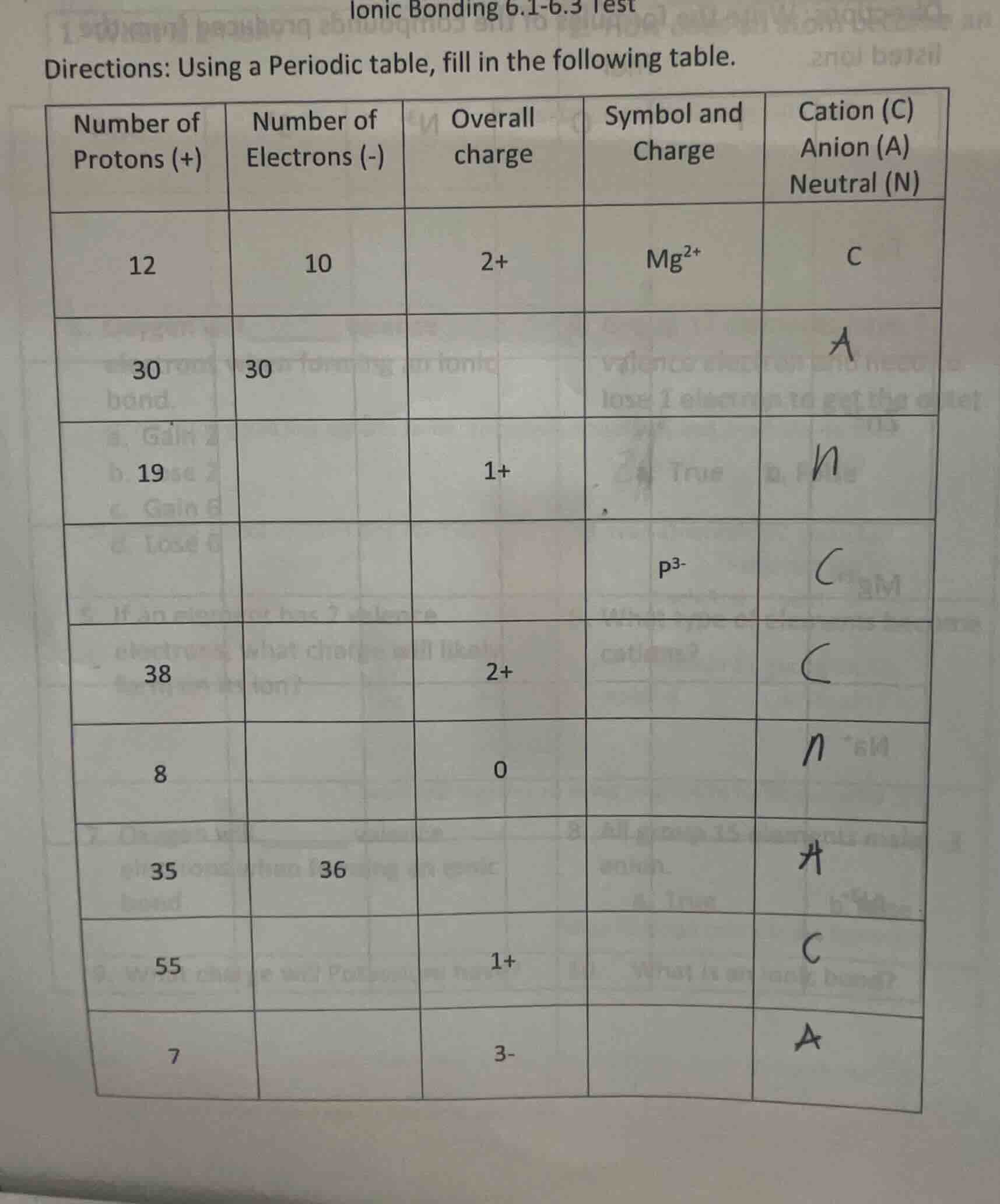
directions: using a periodic table, fill in the following table.
| number of protons (+) | number of electrons (-) | overall charge | symbol and charge | cation (c) anion (a) neutral (n) | ---- | ---- | ---- | ---- | ---- | 12 | 10 | 2+ | $mg^{2+}$ | c | 30 | 30 | a | 19 | 1+ | n | $p^{3-}$ | c | 38 | 2+ | c | 8 | 0 | n | 35 | 36 | a | 55 | 1+ | c | 7 | 3- | a |
Step1: Calculate overall charge
Overall charge = (Number of Protons) - (Number of Electrons)
Step2: Identify element from protons
Element = Periodic table match for proton count
Step3: Write symbol with charge
Symbol = Element symbol + overall charge
Step4: Classify as C/A/N
Cation (C): Charge > 0; Anion (A): Charge < 0; Neutral (N): Charge = 0
---
Row 2 (30 protons, 30 electrons):
Step1: Find overall charge
$\text{Charge} = 30 - 30 = 0$
Step2: Identify element
30 protons = Zinc (Zn)
Step3: Write symbol
$\text{Zn}$
Step4: Classify
Charge = 0 → Neutral (N)
Row 3 (19 protons, charge 1+):
Step1: Find electron count
$\text{Electrons} = 19 - 1 = 18$
Step2: Identify element
19 protons = Potassium (K)
Step3: Write symbol
$\text{K}^+$
Step4: Classify
Charge > 0 → Cation (C)
Row 4 ($\text{P}^{3-}$):
Step1: Find proton count
15 protons = Phosphorus (P)
Step2: Find electron count
$\text{Electrons} = 15 - (-3) = 18$
Step3: Calculate overall charge
$\text{Charge} = 15 - 18 = 3-$
Step4: Classify
Charge < 0 → Anion (A)
Row 5 (38 protons, charge 2+):
Step1: Find electron count
$\text{Electrons} = 38 - 2 = 36$
Step2: Identify element
38 protons = Strontium (Sr)
Step3: Write symbol
$\text{Sr}^{2+}$
Step4: Classify
Charge > 0 → Cation (C)
Row 6 (8 protons, charge 0):
Step1: Find electron count
$\text{Electrons} = 8 - 0 = 8$
Step2: Identify element
8 protons = Oxygen (O)
Step3: Write symbol
$\text{O}$
Step4: Classify
Charge = 0 → Neutral (N)
Row 7 (35 protons, 36 electrons):
Step1: Find overall charge
$\text{Charge} = 35 - 36 = 1-$
Step2: Identify element
35 protons = Bromine (Br)
Step3: Write symbol
$\text{Br}^-$
Step4: Classify
Charge < 0 → Anion (A)
Row 8 (55 protons, charge 1+):
Step1: Find electron count
$\text{Electrons} = 55 - 1 = 54$
Step2: Identify element
55 protons = Cesium (Cs)
Step3: Write symbol
$\text{Cs}^+$
Step4: Classify
Charge > 0 → Cation (C)
Row 9 (7 protons, charge 3-):
Step1: Find electron count
$\text{Electrons} = 7 - (-3) = 10$
Step2: Identify element
7 protons = Nitrogen (N)
Step3: Write symbol
$\text{N}^{3-}$
Step4: Classify
Charge < 0 → Anion (A)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Number of Protons (+) | Number of Electrons (-) | Overall charge | Symbol and Charge | Cation (C) Anion (A) Neutral (N) |
|---|---|---|---|---|
| 30 | 30 | 0 | $\text{Zn}$ | N |
| 19 | 18 | 1+ | $\text{K}^+$ | C |
| 15 | 18 | 3- | $\text{P}^{3-}$ | A |
| 38 | 36 | 2+ | $\text{Sr}^{2+}$ | C |
| 8 | 8 | 0 | $\text{O}$ | N |
| 35 | 36 | 1- | $\text{Br}^-$ | A |
| 55 | 54 | 1+ | $\text{Cs}^+$ | C |
| 7 | 10 | 3- | $\text{N}^{3-}$ | A |