QUESTION IMAGE
Question
le chateliers principle continued
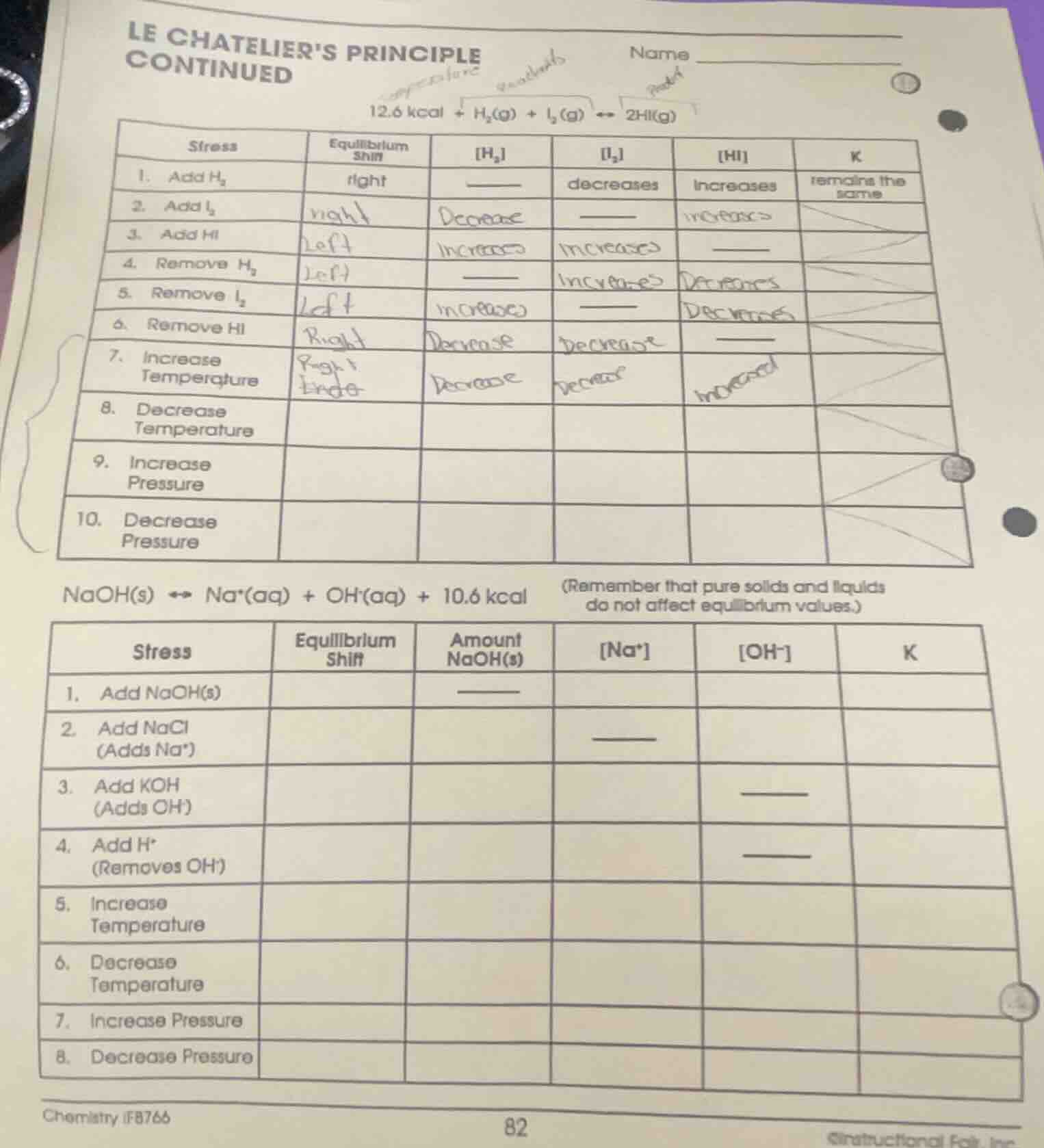
12.6 kcal + h₂(g) + i₂(g) ⇌ 2hi(g)
| stress | equilibrium shift | h₂ | i₂ | hi | k |
|---|---|---|---|---|---|
| 2. add i₂ | ____ | ||||
| 3. add hi | ____ | ||||
| 4. remove h₂ | ____ | ||||
| 5. remove i₂ | ____ | ||||
| 6. remove hi | ____ | ||||
| 7. increase temperature | |||||
| 8. decrease temperature | |||||
| 9. increase pressure | |||||
| 10. decrease pressure |
naoh(s) ⇌ na⁺(aq) + oh⁻(aq) + 10.6 kcal (remember that pure solids and liquids do not affect equilibrium values.)
| stress | equilibrium shift | amount naoh(s) | na⁺ | oh⁻ | k |
|---|---|---|---|---|---|
| 2. add nacl (adds na⁺) | ____ | ||||
| 3. add koh (adds oh⁻) | ____ | ||||
| 4. add h⁺ (removes oh⁻) | ____ | ||||
| 5. increase temperature | |||||
| 6. decrease temperature | |||||
| 7. increase pressure | |||||
| 8. decrease pressure |
chemistry fb766 82
First Equilibrium: \( \boldsymbol{12.6 \text{ kcal} + H_2(g) + I_2(g)
ightleftharpoons 2HI(g)} \)
Stress 1: Add \( H_2 \)
- Equilibrium Shift: Right (given).
- \( [H_2] \): Initially, we add \( H_2 \), but the equilibrium shifts right to consume \( H_2 \) and \( I_2 \) to make \( HI \). However, since we added \( H_2 \), after the shift, \( [H_2] \) will be higher than the original equilibrium but decrease from the moment we added it (as the reaction consumes it). Wait, no—wait, the table says "Equilibrium Shift: right" and \( [I_2] \) decreases, \( [HI] \) increases. For \( [H_2] \): when we add \( H_2 \), the system tries to remove the stress by shifting right, so \( H_2 \) is consumed. But the initial addition increases \( [H_2] \), then the shift decreases it, but the net effect? Wait, the table’s first row: "Add \( H_2 \), Shift: right, \( [I_2] \) decreases, \( [HI] \) increases, \( K \) same." So \( [H_2] \): when we add \( H_2 \), the system shifts right to use \( H_2 \), so \( [H_2] \) decreases from the added amount (but is still higher than original? Wait, no—maybe the table is asking for the change relative to the moment after adding, or the direction. Wait, the first row’s \( [H_2] \) is blank. Let's think: adding \( H_2 \) increases its concentration, then the equilibrium shifts right, consuming \( H_2 \), so \( [H_2] \) decreases (from the peak after addition) but is still higher than original? Wait, no—maybe the table is asking for the trend: when you add \( H_2 \), the system shifts right, so \( [H_2] \) decreases (because the reaction consumes it to make \( HI \)). Wait, maybe the first row’s \( [H_2] \) should be "decreases" (relative to the concentration right after adding \( H_2 \), as the equilibrium shifts to consume it).
Stress 2: Add \( I_2 \)
- Equilibrium Shift: Right (to consume added \( I_2 \) and \( H_2 \) to make \( HI \)).
- \( [H_2] \): Since the shift is right, \( H_2 \) is consumed, so \( [H_2] \) decreases.
- \( [I_2] \): We added \( I_2 \), so initially \( [I_2] \) increases, then the system shifts right to consume \( I_2 \), so \( [I_2] \) decreases from the added amount (but is still higher than original? Wait, the table’s second row: "Add \( I_2 \), Shift: right, \( [H_2] \) (blank), \( [I_2] \) (blank), \( [HI] \) increases." So \( [H_2] \): decreases (because reaction consumes \( H_2 \) to react with added \( I_2 \)). \( [I_2] \): we added \( I_2 \), then the system shifts right to consume it, so \( [I_2] \) decreases (from the added amount) but is still higher than original? Wait, no—maybe the table is asking for the change: when you add \( I_2 \), the system shifts right, so \( [I_2] \) decreases (as it’s consumed), and \( [H_2] \) also decreases (consumed), \( [HI] \) increases.
Stress 3: Add \( HI \)
- Equilibrium Shift: Left (to consume added \( HI \) and make \( H_2 \) and \( I_2 \)).
- \( [HI] \): Adding \( HI \) increases its concentration, then the system shifts left, consuming \( HI \), so \( [HI] \) decreases (from the added amount) as the reaction breaks it into \( H_2 \) and \( I_2 \).
Stress 4: Remove \( H_2 \)
- Equilibrium Shift: Left (to replace removed \( H_2 \), so the reaction goes backward: \( HI \) breaks into \( H_2 \) and \( I_2 \)).
- \( [H_2] \): We removed \( H_2 \), so the system shifts left to make more \( H_2 \), so \( [H_2] \) increases (from the low point after removal) as the reaction produces \( H_2 \).
- \( [I_2] \): Shifting left, \( HI \) breaks into \( H_2 \)…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First Equilibrium: \( \boldsymbol{12.6 \text{ kcal} + H_2(g) + I_2(g)
ightleftharpoons 2HI(g)} \)
Stress 1: Add \( H_2 \)
- Equilibrium Shift: Right (given).
- \( [H_2] \): Initially, we add \( H_2 \), but the equilibrium shifts right to consume \( H_2 \) and \( I_2 \) to make \( HI \). However, since we added \( H_2 \), after the shift, \( [H_2] \) will be higher than the original equilibrium but decrease from the moment we added it (as the reaction consumes it). Wait, no—wait, the table says "Equilibrium Shift: right" and \( [I_2] \) decreases, \( [HI] \) increases. For \( [H_2] \): when we add \( H_2 \), the system tries to remove the stress by shifting right, so \( H_2 \) is consumed. But the initial addition increases \( [H_2] \), then the shift decreases it, but the net effect? Wait, the table’s first row: "Add \( H_2 \), Shift: right, \( [I_2] \) decreases, \( [HI] \) increases, \( K \) same." So \( [H_2] \): when we add \( H_2 \), the system shifts right to use \( H_2 \), so \( [H_2] \) decreases from the added amount (but is still higher than original? Wait, no—maybe the table is asking for the change relative to the moment after adding, or the direction. Wait, the first row’s \( [H_2] \) is blank. Let's think: adding \( H_2 \) increases its concentration, then the equilibrium shifts right, consuming \( H_2 \), so \( [H_2] \) decreases (from the peak after addition) but is still higher than original? Wait, no—maybe the table is asking for the trend: when you add \( H_2 \), the system shifts right, so \( [H_2] \) decreases (because the reaction consumes it to make \( HI \)). Wait, maybe the first row’s \( [H_2] \) should be "decreases" (relative to the concentration right after adding \( H_2 \), as the equilibrium shifts to consume it).
Stress 2: Add \( I_2 \)
- Equilibrium Shift: Right (to consume added \( I_2 \) and \( H_2 \) to make \( HI \)).
- \( [H_2] \): Since the shift is right, \( H_2 \) is consumed, so \( [H_2] \) decreases.
- \( [I_2] \): We added \( I_2 \), so initially \( [I_2] \) increases, then the system shifts right to consume \( I_2 \), so \( [I_2] \) decreases from the added amount (but is still higher than original? Wait, the table’s second row: "Add \( I_2 \), Shift: right, \( [H_2] \) (blank), \( [I_2] \) (blank), \( [HI] \) increases." So \( [H_2] \): decreases (because reaction consumes \( H_2 \) to react with added \( I_2 \)). \( [I_2] \): we added \( I_2 \), then the system shifts right to consume it, so \( [I_2] \) decreases (from the added amount) but is still higher than original? Wait, no—maybe the table is asking for the change: when you add \( I_2 \), the system shifts right, so \( [I_2] \) decreases (as it’s consumed), and \( [H_2] \) also decreases (consumed), \( [HI] \) increases.
Stress 3: Add \( HI \)
- Equilibrium Shift: Left (to consume added \( HI \) and make \( H_2 \) and \( I_2 \)).
- \( [HI] \): Adding \( HI \) increases its concentration, then the system shifts left, consuming \( HI \), so \( [HI] \) decreases (from the added amount) as the reaction breaks it into \( H_2 \) and \( I_2 \).
Stress 4: Remove \( H_2 \)
- Equilibrium Shift: Left (to replace removed \( H_2 \), so the reaction goes backward: \( HI \) breaks into \( H_2 \) and \( I_2 \)).
- \( [H_2] \): We removed \( H_2 \), so the system shifts left to make more \( H_2 \), so \( [H_2] \) increases (from the low point after removal) as the reaction produces \( H_2 \).
- \( [I_2] \): Shifting left, \( HI \) breaks into \( H_2 \) and \( I_2 \), so \( [I_2] \) increases.
- \( [HI] \): Shifting left, \( HI \) is consumed, so \( [HI] \) decreases.
Stress 5: Remove \( I_2 \)
- Equilibrium Shift: Left (to replace removed \( I_2 \), so \( HI \) breaks into \( H_2 \) and \( I_2 \)).
- \( [H_2] \): Shifting left, \( HI \) breaks into \( H_2 \) and \( I_2 \), so \( [H_2] \) increases.
- \( [I_2] \): We removed \( I_2 \), so the system shifts left to make more \( I_2 \), so \( [I_2] \) increases (from the low point after removal) as the reaction produces \( I_2 \).
- \( [HI] \): Shifting left, \( HI \) is consumed, so \( [HI] \) decreases.
Stress 6: Remove \( HI \)
- Equilibrium Shift: Right (to replace removed \( HI \), so \( H_2 \) and \( I_2 \) react to make \( HI \)).
- \( [H_2] \): Shifting right, \( H_2 \) is consumed, so \( [H_2] \) decreases.
- \( [I_2] \): Shifting right, \( I_2 \) is consumed, so \( [I_2] \) decreases.
- \( [HI] \): We removed \( HI \), so the system shifts right to make more \( HI \), so \( [HI] \) increases (from the low point after removal) as the reaction produces \( HI \).
Stress 7: Increase Temperature
- The reaction is endothermic (heat is a reactant: \( 12.6 \text{ kcal} + H_2 + I_2
ightleftharpoons 2HI \)). Increasing temperature (adding heat) favors the endothermic direction (right), so equilibrium shifts right.
- \( [H_2] \): Decreases (consumed to make \( HI \)).
- \( [I_2] \): Decreases (consumed to make \( HI \)).
- \( [HI] \): Increases (produced).
Stress 8: Decrease Temperature
- Decreasing temperature (removing heat) favors the exothermic direction (left, since the reverse reaction is exothermic: \( 2HI
ightleftharpoons H_2 + I_2 + 12.6 \text{ kcal} \)). So equilibrium shifts left.
- \( [H_2] \): Increases (produced from \( HI \) decomposition).
- \( [I_2] \): Increases (produced from \( HI \) decomposition).
- \( [HI] \): Decreases (consumed in decomposition).
Stress 9: Increase Pressure
- The reaction has \( 2 \) moles of gas on the right (\( 2HI \)) and \( 2 \) moles on the left (\( H_2 + I_2 \)) (since \( H_2 \) and \( I_2 \) are each 1 mole, total 2 moles left; \( HI \) is 2 moles right). So \( \Delta n = 0 \) (moles of gas: left = 2, right = 2). Therefore, pressure changes have no effect on equilibrium (Le Chatelier’s principle: pressure affects equilibrium only when \( \Delta n
eq 0 \)). So equilibrium shift: no shift.
- \( [H_2] \), \( [I_2] \), \( [HI] \): No change (since equilibrium doesn’t shift).
- \( K \): Remains same (temperature is constant, \( K \) depends only on temperature).
Stress 10: Decrease Pressure
- Same as increasing pressure: \( \Delta n = 0 \), so no effect. Equilibrium shift: no shift.
- \( [H_2] \), \( [I_2] \), \( [HI] \): No change.
Second Equilibrium: \( \boldsymbol{NaOH(s)
ightleftharpoons Na^+(aq) + OH^-(aq) + 10.6 \text{ kcal}} \) (exothermic, heat is a product)
Stress 1: Add \( NaOH(s) \)
- \( NaOH \) is a solid. Pure solids/liquids do not affect equilibrium (their concentration is constant). So adding \( NaOH(s) \) has no effect.
- Equilibrium Shift: No shift.
- Amount \( NaOH(s) \): Increases (we added it), but since it’s a solid, the equilibrium doesn’t shift, so the amount of \( NaOH(s) \) remains the same (wait, no—adding solid \( NaOH \) increases its amount, but the equilibrium doesn’t shift because solids don’t affect \( Q \) or \( K \). So "Amount \( NaOH(s) \)": increases (because we added it), but the equilibrium doesn’t shift, so \( [Na^+] \), \( [OH^-] \), \( K \) remain same.
Stress 2: Add \( NaCl \) (adds \( Na^+ \))
- Adding \( Na^+ \) (a product) increases \( [Na^+] \), so the system shifts left (to remove the excess \( Na^+ \) by making more \( NaOH(s) \)).
- Equilibrium Shift: Left.
- Amount \( NaOH(s) \): Increases (since the reaction shifts left, producing \( NaOH(s) \)).
- \( [Na^+] \): Initially increases (from \( NaCl \) addition), then the system shifts left, consuming \( Na^+ \), so \( [Na^+] \) decreases (from the peak after addition) but is still higher than original? Wait, the table asks for the change: adding \( Na^+ \) increases \( [Na^+] \), then the shift decreases it, but the net effect? The table’s second row: "Add \( NaCl \) (adds \( Na^+ \))", so \( [Na^+] \) initially increases, then the system shifts left to consume \( Na^+ \), so \( [Na^+] \) decreases (relative to the added amount, but is still higher than original? Or the trend: when you add \( Na^+ \), the system shifts left, so \( [Na^+] \) decreases (from the added concentration) as the reaction consumes it to make \( NaOH(s) \)).
Stress 3: Add \( KOH \) (adds \( OH^- \))
- Adding \( OH^- \) (a product) increases \( [OH^-] \), so the system shifts left (to remove excess \( OH^- \) by making \( NaOH(s) \)).
- Equilibrium Shift: Left.
- Amount \( NaOH(s) \): Increases (produced as the reaction shifts left).
- \( [OH^-] \): Initially increases (from \( KOH \) addition), then the system shifts left, consuming \( OH^- \), so \( [OH^-] \) decreases (from the peak after addition) but is still higher than original?
Stress 4: Add \( H^+ \) (removes \( OH^- \) via \( H^+ + OH^-
ightarrow H_2O \))
- Removing \( OH^- \) (a product) decreases \( [OH^-] \), so the system shifts right (to replace the removed \( OH^- \) by dissolving \( NaOH(s) \) into \( Na^+ \) and \( OH^- \)).
- Equilibrium Shift: Right.
- Amount \( NaOH(s) \): Decreases (consumed as the reaction shifts right to make \( Na^+ \) and \( OH^- \)).
- \( [OH^-] \): Initially decreases (from \( H^+ \) addition), then the system shifts right, producing \( OH^- \), so \( [OH^-] \) increases (from the low point after removal) as the reaction produces \( OH^- \).
Stress 5: Increase Temperature
- The reaction is exothermic (heat is a product: \( NaOH(s)
ightleftharpoons Na^+ + OH^- + \text{heat} \)). Increasing temperature (adding heat) is like adding a product, so the system shifts left (to remove the excess heat by favoring the endothermic direction, which is the reverse reaction: \( Na^+ + OH^- + \text{heat}
ightarrow NaOH(s) \)).
- Equilibrium Shift: Left.
- Amount \( NaOH(s) \): Increases (produced as the reaction shifts left).
- \( [Na^+] \), \( [OH^-] \): Decrease (consumed as the reaction shifts left to make \( NaOH(s) \)).
Stress 6: Decrease Temperature
- Decreasing temperature (removing heat) is like removing a product, so the system shifts right (to replace the removed heat by favoring the exothermic direction, which is the forward reaction: \( NaOH(s)
ightleftharpoons Na^+ + OH^- + \text{heat} \)).
- Equilibrium Shift: Right.
- Amount \( NaOH(s) \): Decreases (consumed as the reaction shifts right to make \( Na^+ \) and \( OH^- \)).
- \( [Na^+] \), \( [OH^-] \): Increase (produced as the reaction shifts right).
Stress 7: Increase Pressure
- \( NaOH(s) \) is a solid, \( Na^+ \) and \( OH^- \) are aqueous. Pressure changes have negligible effect on aqueous/solid equilibria (since pressure affects gases[SSE onError error]