QUESTION IMAGE
Question
learning: molecules and compounds calculations
question
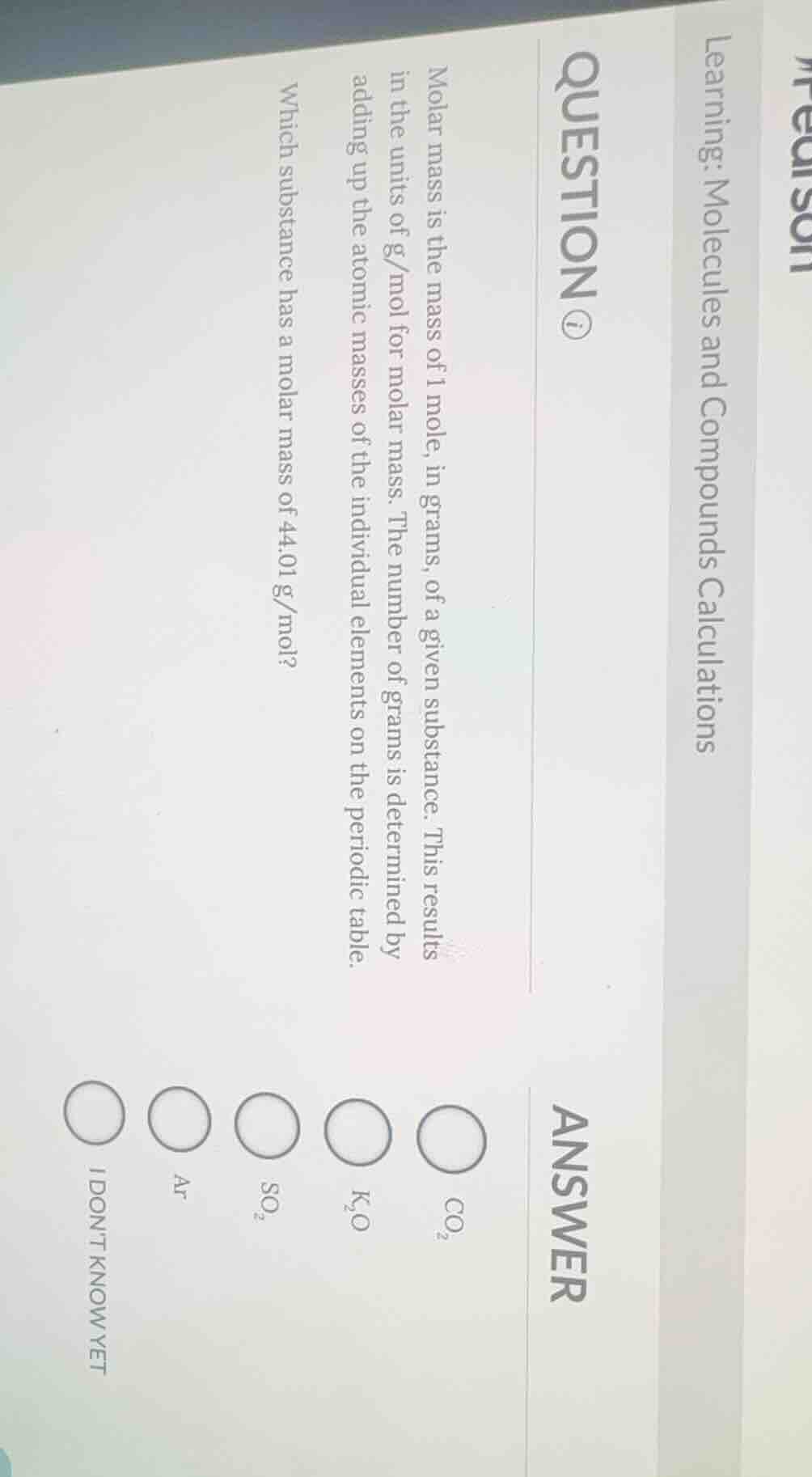
molar mass is the mass of 1 mole, in grams, of a given substance. this results in the units of g/mol for molar mass. the number of grams is determined by adding up the atomic masses of the individual elements on the periodic table.
which substance has a molar mass of 44.01 g/mol?
answer
$co_2$
$k_2o$
$so_2$
ar
i dont know yet
Step1: Find atomic masses (periodic table)
Atomic masses: C = 12.01 g/mol, O = 16.00 g/mol, K = 39.10 g/mol, S = 32.07 g/mol, Ar = 39.95 g/mol
Step2: Calculate molar mass of $\text{CO}_2$
$\text{Molar mass of }\text{CO}_2 = 12.01 + 2\times16.00 = 44.01\text{ g/mol}$
Step3: Verify other options
$\text{Molar mass of }\text{K}_2\text{O} = 2\times39.10 + 16.00 = 94.20\text{ g/mol}$
$\text{Molar mass of }\text{SO}_2 = 32.07 + 2\times16.00 = 64.07\text{ g/mol}$
$\text{Molar mass of Ar} = 39.95\text{ g/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\text{CO}_2$