QUESTION IMAGE
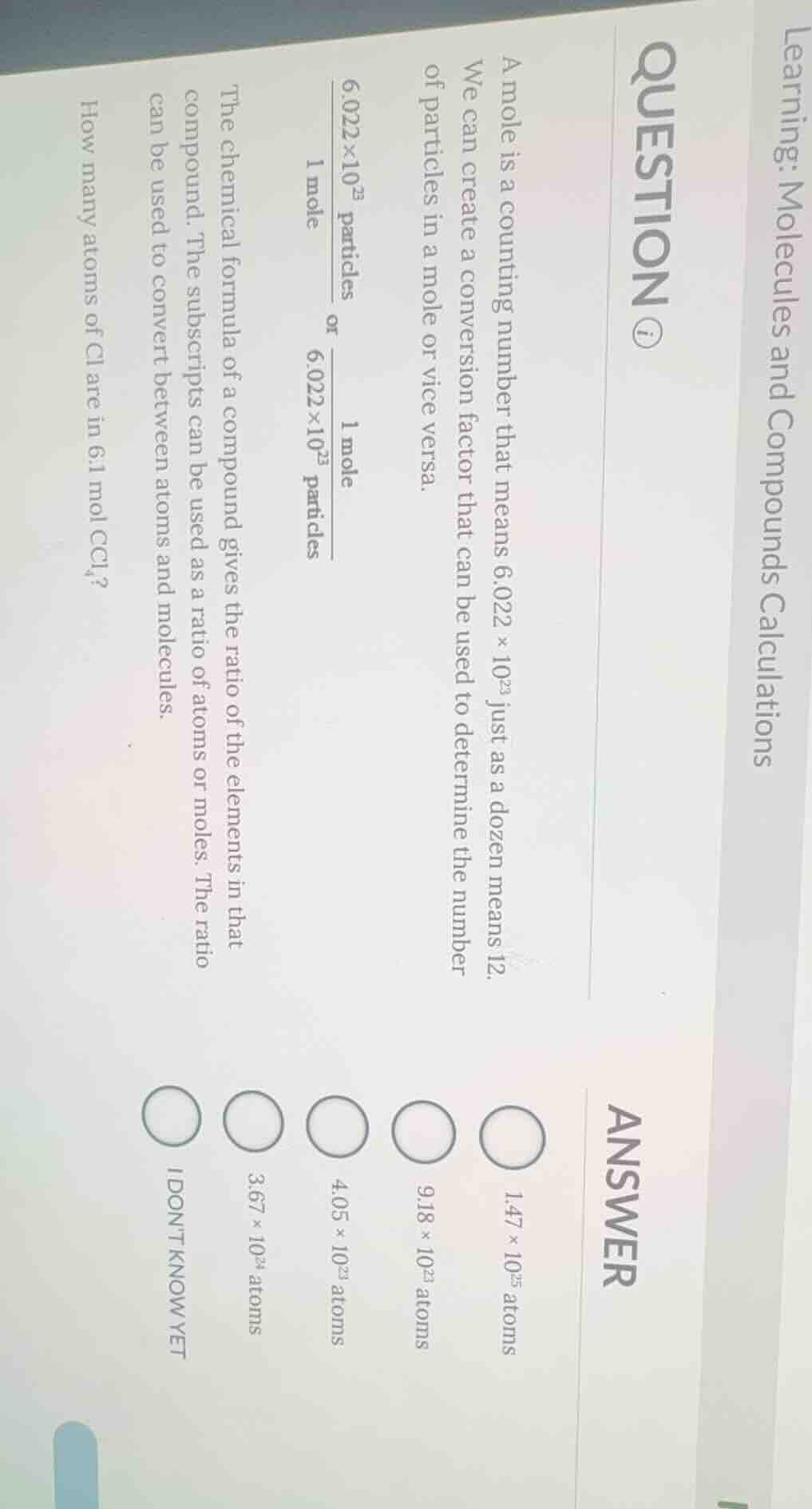
Question
learning: molecules and compounds calculations
question ⓘ
a mole is a counting number that means $6.022 \times 10^{23}$ just as a dozen means 12.
we can create a conversion factor that can be used to determine the number
of particles in a mole or vice versa.
$\frac{6.022 \times 10^{23}\text{ particles}}{1\text{ mole}}$ or $\frac{1\text{ mole}}{6.022 \times 10^{23}\text{ particles}}$
the chemical formula of a compound gives the ratio of the elements in that
compound. the subscripts can be used as a ratio of atoms or moles. the ratio
can be used to convert between atoms and molecules.
how many atoms of cl are in $6.1\text{ mol }ccl_4$?
answer
$1.47 \times 10^{25}$ atoms
$9.18 \times 10^{23}$ atoms
$4.05 \times 10^{23}$ atoms
$3.67 \times 10^{24}$ atoms
i dont know yet
Step1: Find moles of Cl from $\text{CCl}_4$
1 mole of $\text{CCl}_4$ contains 4 moles of Cl.
Moles of Cl = $6.1\ \text{mol}\ \text{CCl}_4 \times 4 = 24.4\ \text{mol Cl}$
Step2: Convert moles to atoms
Use Avogadro's number: $6.022 \times 10^{23}$ atoms/mol.
Number of Cl atoms = $24.4\ \text{mol} \times 6.022 \times 10^{23}\ \text{atoms/mol}$
Step3: Calculate final value
$24.4 \times 6.022 \times 10^{23} = 147.0368 \times 10^{23} = 1.47 \times 10^{25}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.47 × 10²⁵ atoms