QUESTION IMAGE
Question
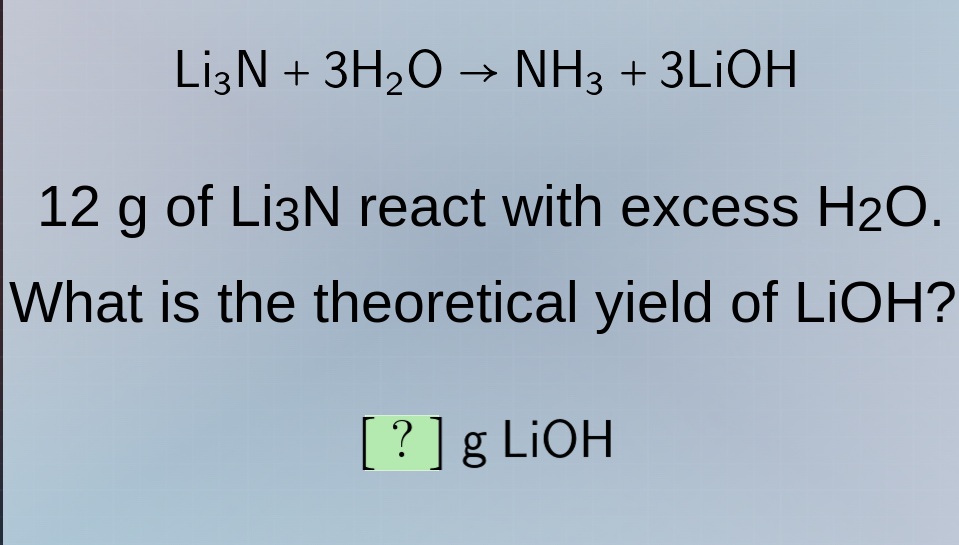
li₃n + 3h₂o → nh₃ + 3lioh
12 g of li₃n react with excess h₂o.
what is the theoretical yield of lioh?
? g lioh
Step1: Calculate molar mass of \( \text{Li}_3\text{N} \)
Molar mass of \( \text{Li} = 6.94 \, \text{g/mol} \), \( \text{N} = 14.01 \, \text{g/mol} \)
\( \text{Molar mass of } \text{Li}_3\text{N} = 3\times6.94 + 14.01 = 20.82 + 14.01 = 34.83 \, \text{g/mol} \)
Step2: Calculate moles of \( \text{Li}_3\text{N} \)
Given mass of \( \text{Li}_3\text{N} = 12 \, \text{g} \)
\( \text{Moles of } \text{Li}_3\text{N} = \frac{\text{Given mass}}{\text{Molar mass}} = \frac{12}{34.83} \approx 0.3445 \, \text{mol} \)
Step3: Use stoichiometry to find moles of \( \text{LiOH} \)
From the balanced equation \( \text{Li}_3\text{N} + 3\text{H}_2\text{O}
ightarrow \text{NH}_3 + 3\text{LiOH} \), 1 mole of \( \text{Li}_3\text{N} \) produces 3 moles of \( \text{LiOH} \)
\( \text{Moles of } \text{LiOH} = 3 \times \text{Moles of } \text{Li}_3\text{N} = 3 \times 0.3445 \approx 1.0335 \, \text{mol} \)
Step4: Calculate molar mass of \( \text{LiOH} \)
Molar mass of \( \text{Li} = 6.94 \, \text{g/mol} \), \( \text{O} = 16.00 \, \text{g/mol} \), \( \text{H} = 1.01 \, \text{g/mol} \)
\( \text{Molar mass of } \text{LiOH} = 6.94 + 16.00 + 1.01 = 23.95 \, \text{g/mol} \)
Step5: Calculate mass of \( \text{LiOH} \)
\( \text{Mass of } \text{LiOH} = \text{Moles} \times \text{Molar mass} = 1.0335 \times 23.95 \approx 24.76 \, \text{g} \) (Rounding to appropriate significant figures or as needed, but let's check the calculation again for precision)
Wait, let's recalculate step by step with more precision:
Step1: Molar mass of \( \text{Li}_3\text{N} \): \( 3\times6.941 + 14.0067 = 20.823 + 14.0067 = 34.8297 \, \text{g/mol} \)
Step2: Moles of \( \text{Li}_3\text{N} \): \( \frac{12}{34.8297} \approx 0.3445 \, \text{mol} \) (exact value: \( 12 \div 34.8297 \approx 0.3445 \))
Step3: Moles of \( \text{LiOH} \): \( 0.3445 \times 3 = 1.0335 \, \text{mol} \)
Step4: Molar mass of \( \text{LiOH} \): \( 6.941 + 16.00 + 1.008 = 23.949 \, \text{g/mol} \) (using more precise atomic masses: Li=6.941, O=16.00, H=1.008)
Step5: Mass of \( \text{LiOH} \): \( 1.0335 \times 23.949 \approx 1.0335 \times 23.95 \approx 24.76 \, \text{g} \). But let's do it more accurately:
\( 1.0335 \times 23.949 = (1 + 0.0335) \times 23.949 = 23.949 + 0.0335 \times 23.949 \approx 23.949 + 0.802 = 24.751 \, \text{g} \approx 24.8 \, \text{g} \) (or maybe the answer is expected to be around 24.7 or 24.8 g. Let's check with exact calculation:
\( 12 \, \text{g Li}_3\text{N} \times \frac{1 \, \text{mol Li}_3\text{N}}{34.83 \, \text{g Li}_3\text{N}} \times \frac{3 \, \text{mol LiOH}}{1 \, \text{mol Li}_3\text{N}} \times \frac{23.95 \, \text{g LiOH}}{1 \, \text{mol LiOH}} \)
Calculate step by step:
First, \( 12 \div 34.83 = 0.3445 \)
Then, \( 0.3445 \times 3 = 1.0335 \)
Then, \( 1.0335 \times 23.95 = 24.75 \, \text{g} \) (approx 24.8 g)
Alternatively, using exact fractions:
\( \frac{12 \times 3 \times 23.95}{34.83} = \frac{12 \times 71.85}{34.83} = \frac{862.2}{34.83} \approx 24.75 \, \text{g} \)
So the theoretical yield of \( \text{LiOH} \) is approximately \( 24.8 \, \text{g} \) (or more precisely around 24.7 - 24.8 g)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \approx 24.8 \) (or more accurately \( 24.7 - 24.8 \) g, but the precise calculation gives approximately 24.8 g)