QUESTION IMAGE
Question
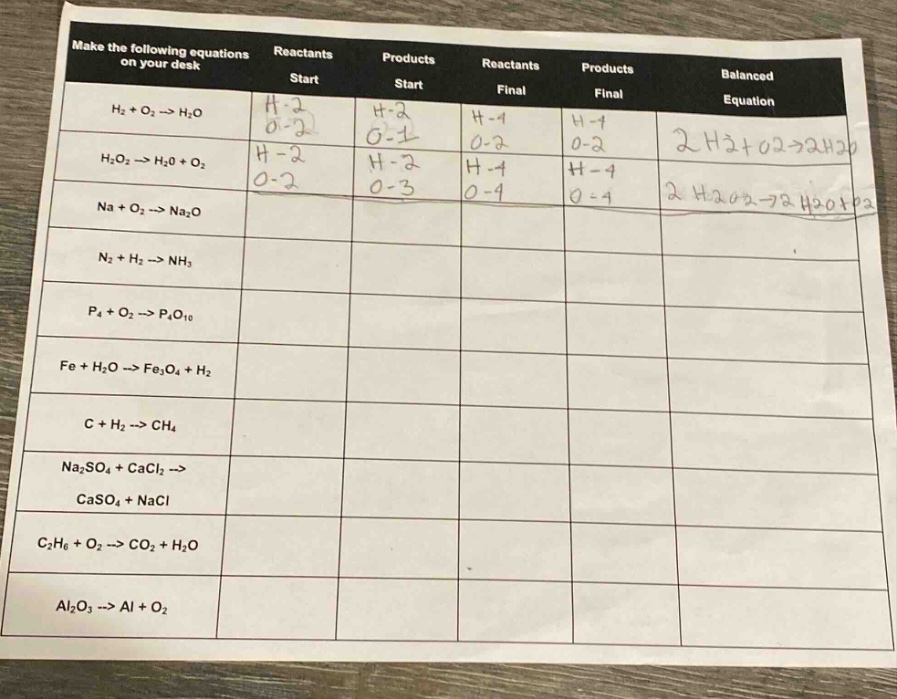
make the following equations on your desk
| reactants start | products start | reactants final | products final | balanced equation | |
|---|---|---|---|---|---|
| $ce{h_{2}o_{2} -> h_{2}o + o_{2}}$ | |||||
| $ce{na + o_{2} -> na_{2}o}$ | |||||
| $ce{n_{2} + h_{2} -> nh_{3}}$ | |||||
| $ce{p_{4} + o_{2} -> p_{4}o_{10}}$ | |||||
| $ce{fe + h_{2}o -> fe_{3}o_{4} + h_{2}}$ | |||||
| $ce{c + h_{2} -> ch_{4}}$ | |||||
| $ce{na_{2}so_{4} + cacl_{2} -> caso_{4} + nacl}$ | |||||
| $ce{c_{2}h_{6} + o_{2} -> co_{2} + h_{2}o}$ | |||||
| $ce{al_{2}o_{3} -> al + o_{2}}$ |
To solve these chemical equations, we need to balance them by ensuring the number of each type of atom is the same on both sides of the equation. Here are the balanced equations for each:
1. $\boldsymbol{H_2 + O_2
ightarrow H_2O}$
Step 1: Count atoms
- Reactants: $H = 2$, $O = 2$
- Products: $H = 2$, $O = 1$
Step 2: Balance O
Multiply $H_2O$ by 2: $H_2 + O_2
ightarrow 2H_2O$
Now, $H$ on products: $4$
Step 3: Balance H
Multiply $H_2$ by 2: $2H_2 + O_2
ightarrow 2H_2O$
Now, $H = 4$ (both sides), $O = 2$ (both sides)
Balanced Equation: $2H_2 + O_2 = 2H_2O$
2. $\boldsymbol{H_2O_2
ightarrow H_2O + O_2}$
Step 1: Count atoms
- Reactants: $H = 2$, $O = 2$
- Products: $H = 2$, $O = 3$ (1 in $H_2O$, 2 in $O_2$)
Step 2: Balance O
Multiply $H_2O_2$ by 2: $2H_2O_2
ightarrow H_2O + O_2$
Now, $H = 4$, $O = 4$
Step 3: Balance H and O
Multiply $H_2O$ by 2: $2H_2O_2
ightarrow 2H_2O + O_2$
Now, $H = 4$ (both sides), $O = 4$ (both sides)
Balanced Equation: $2H_2O_2 = 2H_2O + O_2$
3. $\boldsymbol{Na + O_2
ightarrow Na_2O}$
Step 1: Count atoms
- Reactants: $Na = 1$, $O = 2$
- Products: $Na = 2$, $O = 1$
Step 2: Balance O
Multiply $Na_2O$ by 2: $Na + O_2
ightarrow 2Na_2O$
Now, $O = 2$ (both sides), $Na = 4$
Step 3: Balance Na
Multiply $Na$ by 4: $4Na + O_2
ightarrow 2Na_2O$
Now, $Na = 4$ (both sides), $O = 2$ (both sides)
Balanced Equation: $4Na + O_2 = 2Na_2O$
4. $\boldsymbol{N_2 + H_2
ightarrow NH_3}$
Step 1: Count atoms
- Reactants: $N = 2$, $H = 2$
- Products: $N = 1$, $H = 3$
Step 2: Balance N
Multiply $NH_3$ by 2: $N_2 + H_2
ightarrow 2NH_3$
Now, $N = 2$ (both sides), $H = 6$
Step 3: Balance H
Multiply $H_2$ by 3: $N_2 + 3H_2
ightarrow 2NH_3$
Now, $N = 2$, $H = 6$ (both sides)
Balanced Equation: $N_2 + 3H_2 = 2NH_3$
5. $\boldsymbol{P_4 + O_2
ightarrow P_4O_{10}}$
Step 1: Count atoms
- Reactants: $P = 4$, $O = 2$
- Products: $P = 4$, $O = 10$
Step 2: Balance O
Multiply $O_2$ by 5: $P_4 + 5O_2
ightarrow P_4O_{10}$
Now, $O = 10$ (both sides), $P = 4$ (both sides)
Balanced Equation: $P_4 + 5O_2 = P_4O_{10}$
6. $\boldsymbol{Fe + H_2O
ightarrow Fe_3O_4 + H_2}$
Step 1: Count atoms
- Reactants: $Fe = 1$, $H = 2$, $O = 1$
- Products: $Fe = 3$, $H = 2$, $O = 4$
Step 2: Balance Fe
Multiply $Fe$ by 3: $3Fe + H_2O
ightarrow Fe_3O_4 + H_2$
Now, $Fe = 3$ (both sides), $O = 1$ (reactants), $O = 4$ (products)
Step 3: Balance O
Multiply $H_2O$ by 4: $3Fe + 4H_2O
ightarrow Fe_3O_4 + H_2$
Now, $O = 4$ (both sides), $H = 8$ (reactants)
Step 4: Balance H
Multiply $H_2$ by 4: $3Fe + 4H_2O = Fe_3O_4 + 4H_2$
Now, $H = 8$ (both sides), $Fe = 3$, $O = 4$ (both sides)
Balanced Equation: $3Fe + 4H_2O = Fe_3O_4 + 4H_2$
7. $\boldsymbol{C + H_2
ightarrow CH_4}$
Step 1: Count atoms
- Reactants: $C = 1$, $H = 2$
- Products: $C = 1$, $H = 4$
Step 2: Balance H
Multiply $H_2$ by 2: $C + 2H_2 = CH_4$
Now, $H = 4$ (both sides), $C = 1$ (both sides)
Balanced Equation: $C + 2H_2 = CH_4$
8. $\boldsymbol{Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + NaCl}$
Step 1: Count atoms
- Reactants: $Na = 2$, $S = 1$, $O = 4$, $Ca = 1$, $Cl = 2$
- Products: $Ca = 1$, $S = 1$, $O = 4$, $Na = 1$, $Cl = 1$
Step 2: Balance Na and Cl
Multiply $NaCl$ by 2: $Na_2SO_4 + CaCl_2 = CaSO_4 + 2NaCl$
Now, $Na = 2$ (both sides), $Cl = 2$ (both sides), others balanced
Balanced Equation: $Na_2SO_4 + CaCl_2 = CaSO_4 + 2NaCl$
9. $\boldsymbol{C_2H_6 + O_2
ightarrow CO_2 + H_2O}$
Step 1: Count atoms
- Reactants: $C = 2$, $H = 6$, $O = 2$
- Products: $C = 1$, $H = 2$, $O = 3$
Step 2: Balance C
Multiply…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve these chemical equations, we need to balance them by ensuring the number of each type of atom is the same on both sides of the equation. Here are the balanced equations for each:
1. $\boldsymbol{H_2 + O_2
ightarrow H_2O}$
Step 1: Count atoms
- Reactants: $H = 2$, $O = 2$
- Products: $H = 2$, $O = 1$
Step 2: Balance O
Multiply $H_2O$ by 2: $H_2 + O_2
ightarrow 2H_2O$
Now, $H$ on products: $4$
Step 3: Balance H
Multiply $H_2$ by 2: $2H_2 + O_2
ightarrow 2H_2O$
Now, $H = 4$ (both sides), $O = 2$ (both sides)
Balanced Equation: $2H_2 + O_2 = 2H_2O$
2. $\boldsymbol{H_2O_2
ightarrow H_2O + O_2}$
Step 1: Count atoms
- Reactants: $H = 2$, $O = 2$
- Products: $H = 2$, $O = 3$ (1 in $H_2O$, 2 in $O_2$)
Step 2: Balance O
Multiply $H_2O_2$ by 2: $2H_2O_2
ightarrow H_2O + O_2$
Now, $H = 4$, $O = 4$
Step 3: Balance H and O
Multiply $H_2O$ by 2: $2H_2O_2
ightarrow 2H_2O + O_2$
Now, $H = 4$ (both sides), $O = 4$ (both sides)
Balanced Equation: $2H_2O_2 = 2H_2O + O_2$
3. $\boldsymbol{Na + O_2
ightarrow Na_2O}$
Step 1: Count atoms
- Reactants: $Na = 1$, $O = 2$
- Products: $Na = 2$, $O = 1$
Step 2: Balance O
Multiply $Na_2O$ by 2: $Na + O_2
ightarrow 2Na_2O$
Now, $O = 2$ (both sides), $Na = 4$
Step 3: Balance Na
Multiply $Na$ by 4: $4Na + O_2
ightarrow 2Na_2O$
Now, $Na = 4$ (both sides), $O = 2$ (both sides)
Balanced Equation: $4Na + O_2 = 2Na_2O$
4. $\boldsymbol{N_2 + H_2
ightarrow NH_3}$
Step 1: Count atoms
- Reactants: $N = 2$, $H = 2$
- Products: $N = 1$, $H = 3$
Step 2: Balance N
Multiply $NH_3$ by 2: $N_2 + H_2
ightarrow 2NH_3$
Now, $N = 2$ (both sides), $H = 6$
Step 3: Balance H
Multiply $H_2$ by 3: $N_2 + 3H_2
ightarrow 2NH_3$
Now, $N = 2$, $H = 6$ (both sides)
Balanced Equation: $N_2 + 3H_2 = 2NH_3$
5. $\boldsymbol{P_4 + O_2
ightarrow P_4O_{10}}$
Step 1: Count atoms
- Reactants: $P = 4$, $O = 2$
- Products: $P = 4$, $O = 10$
Step 2: Balance O
Multiply $O_2$ by 5: $P_4 + 5O_2
ightarrow P_4O_{10}$
Now, $O = 10$ (both sides), $P = 4$ (both sides)
Balanced Equation: $P_4 + 5O_2 = P_4O_{10}$
6. $\boldsymbol{Fe + H_2O
ightarrow Fe_3O_4 + H_2}$
Step 1: Count atoms
- Reactants: $Fe = 1$, $H = 2$, $O = 1$
- Products: $Fe = 3$, $H = 2$, $O = 4$
Step 2: Balance Fe
Multiply $Fe$ by 3: $3Fe + H_2O
ightarrow Fe_3O_4 + H_2$
Now, $Fe = 3$ (both sides), $O = 1$ (reactants), $O = 4$ (products)
Step 3: Balance O
Multiply $H_2O$ by 4: $3Fe + 4H_2O
ightarrow Fe_3O_4 + H_2$
Now, $O = 4$ (both sides), $H = 8$ (reactants)
Step 4: Balance H
Multiply $H_2$ by 4: $3Fe + 4H_2O = Fe_3O_4 + 4H_2$
Now, $H = 8$ (both sides), $Fe = 3$, $O = 4$ (both sides)
Balanced Equation: $3Fe + 4H_2O = Fe_3O_4 + 4H_2$
7. $\boldsymbol{C + H_2
ightarrow CH_4}$
Step 1: Count atoms
- Reactants: $C = 1$, $H = 2$
- Products: $C = 1$, $H = 4$
Step 2: Balance H
Multiply $H_2$ by 2: $C + 2H_2 = CH_4$
Now, $H = 4$ (both sides), $C = 1$ (both sides)
Balanced Equation: $C + 2H_2 = CH_4$
8. $\boldsymbol{Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + NaCl}$
Step 1: Count atoms
- Reactants: $Na = 2$, $S = 1$, $O = 4$, $Ca = 1$, $Cl = 2$
- Products: $Ca = 1$, $S = 1$, $O = 4$, $Na = 1$, $Cl = 1$
Step 2: Balance Na and Cl
Multiply $NaCl$ by 2: $Na_2SO_4 + CaCl_2 = CaSO_4 + 2NaCl$
Now, $Na = 2$ (both sides), $Cl = 2$ (both sides), others balanced
Balanced Equation: $Na_2SO_4 + CaCl_2 = CaSO_4 + 2NaCl$
9. $\boldsymbol{C_2H_6 + O_2
ightarrow CO_2 + H_2O}$
Step 1: Count atoms
- Reactants: $C = 2$, $H = 6$, $O = 2$
- Products: $C = 1$, $H = 2$, $O = 3$
Step 2: Balance C
Multiply $CO_2$ by 2: $C_2H_6 + O_2
ightarrow 2CO_2 + H_2O$
Now, $C = 2$ (both sides), $H = 6$ (reactants), $H = 2$ (products)
Step 3: Balance H
Multiply $H_2O$ by 3: $C_2H_6 + O_2
ightarrow 2CO_2 + 3H_2O$
Now, $H = 6$ (both sides), $O = 7$ (products: $4 + 3 = 7$)
Step 4: Balance O
Multiply $O_2$ by $\frac{7}{2}$: $C_2H_6 + \frac{7}{2}O_2
ightarrow 2CO_2 + 3H_2O$
To eliminate fraction, multiply all by 2: $2C_2H_6 + 7O_2 = 4CO_2 + 6H_2O$
Now, $C = 4$, $H = 12$, $O = 14$ (both sides)
Balanced Equation: $2C_2H_6 + 7O_2 = 4CO_2 + 6H_2O$
10. $\boldsymbol{Al_2O_3
ightarrow Al + O_2}$
Step 1: Count atoms
- Reactants: $Al = 2$, $O = 3$
- Products: $Al = 1$, $O = 2$
Step 2: Balance Al
Multiply $Al$ by 2: $Al_2O_3
ightarrow 2Al + O_2$
Now, $Al = 2$ (both sides), $O = 3$ (reactants), $O = 2$ (products)
Step 3: Balance O
Multiply $Al_2O_3$ by 2 and $O_2$ by 3: $2Al_2O_3
ightarrow 4Al + 3O_2$
Now, $Al = 4$ (both sides), $O = 6$ (both sides)
Balanced Equation: $2Al_2O_3 = 4Al + 3O_2$
Summary of Balanced Equations:
- $2H_2 + O_2 = 2H_2O$
- $2H_2O_2 = 2H_2O + O_2$
- $4Na + O_2 = 2Na_2O$
- $N_2 + 3H_2 = 2NH_3$
- $P_4 + 5O_2 = P_4O_{10}$
- $3Fe + 4H_2O = Fe_3O_4 + 4H_2$
- $C + 2H_2 = CH_4$
- $Na_2SO_4 + CaCl_2 = CaSO_4 + 2NaCl$
- $2C_2H_6 + 7O_2 = 4CO_2 + 6H_2O$
- $2Al_2O_3 = 4Al + 3O_2$