QUESTION IMAGE
Question
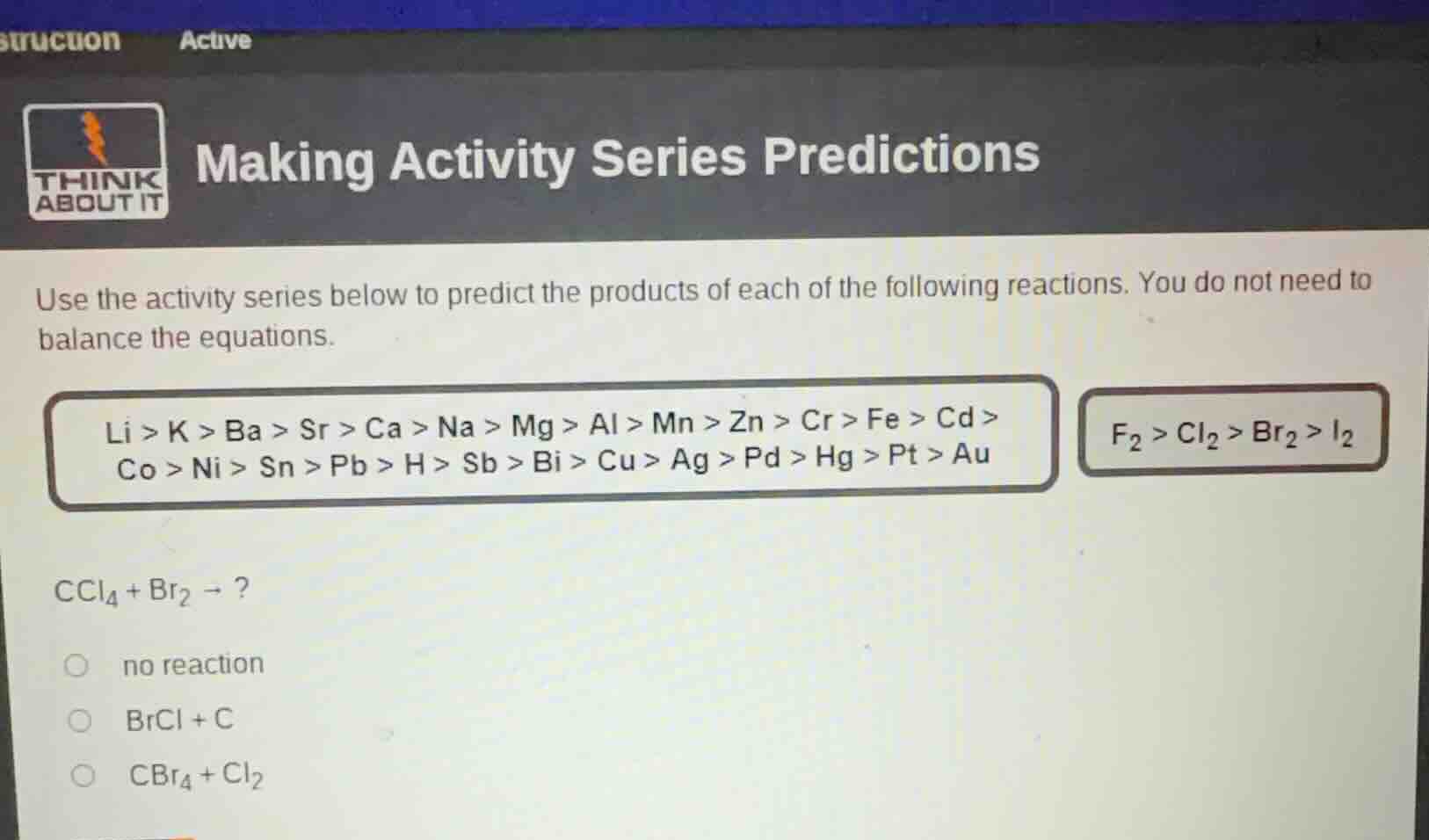
making activity series predictions
use the activity series below to predict the products of each of the following reactions. you do not need to balance the equations.
li > k > ba > sr > ca > na > mg > al > mn > zn > cr > fe > cd >
co > ni > sn > pb > h > sb > bi > cu > ag > pd > hg > pt > au
$f_{2}>cl_{2}>br_{2}>i_{2}$
$ccl_{4}+br_{2} \to ?$
no reaction
$brcl + c$
$cbr_{4}+cl_{2}$
Step1: Compare halogen reactivity
From the given series: $\text{Cl}_2 > \text{Br}_2$
Step2: Determine reaction feasibility
A less reactive halogen ($\text{Br}_2$) cannot displace a more reactive halogen ($\text{Cl}$) from its compound ($\text{CCl}_4$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
no reaction