QUESTION IMAGE
Question
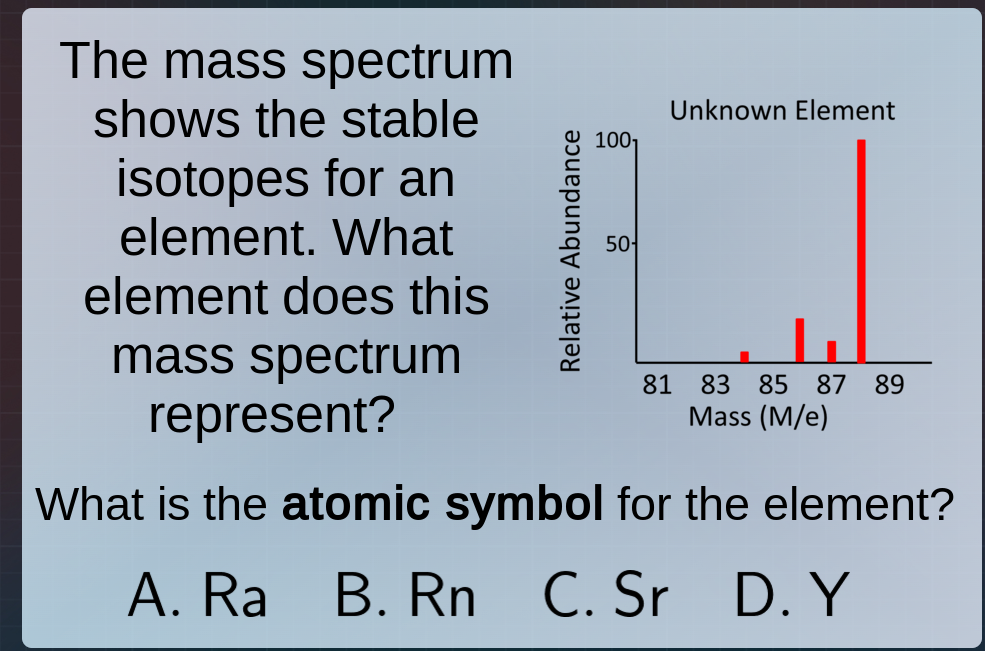
the mass spectrum shows the stable isotopes for an element. what element does this mass spectrum represent? what is the atomic symbol for the element? a. ra b. rn c. sr d. y (with a mass spectrum chart for unknown element, x - axis: mass (m/e) with values 81, 83, 85, 87, 89; y - axis: relative abundance with values 50, 100)
Brief Explanations
To determine the element, we analyze the mass spectrum and atomic properties:
- Isotope Masses and Abundance: The mass spectrum shows isotopes with masses around 83, 85, 87, 89 (with 89 being most abundant). We calculate the average atomic mass (weighted by abundance, though the dominant isotope here is ~89, and we check atomic masses of options:
- Ra (Radium): Atomic mass ~226 (too high).
- Rn (Radon): Atomic mass ~222 (too high).
- Sr (Strontium): Has stable isotopes including Sr - 84, Sr - 86, Sr - 87, Sr - 88, Sr - 89 (masses align with the spectrum’s ~83 - 89 range, and Sr’s isotopes match the mass/abundance pattern here).
- Y (Yttrium): Atomic mass ~89, but Y has fewer stable isotopes in this range, and Sr’s isotope distribution better matches the spectrum’s multiple isotopes (83, 85, 87, 89 - Sr has isotopes in this mass range with varying abundances).
- Atomic Mass Match: Strontium (Sr) has stable isotopes with masses close to 83, 85, 87, 89 (e.g., Sr - 84, Sr - 86, Sr - 87, Sr - 88, Sr - 89), and the dominant isotope here (89) aligns with Sr - 89’s existence. Ra and Rn have much higher atomic masses (radioactive, heavy elements), and Y has a single dominant isotope ~89 but fewer isotopes in the 83 - 87 range. Thus, Sr (option C) is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Sr