QUESTION IMAGE
Question
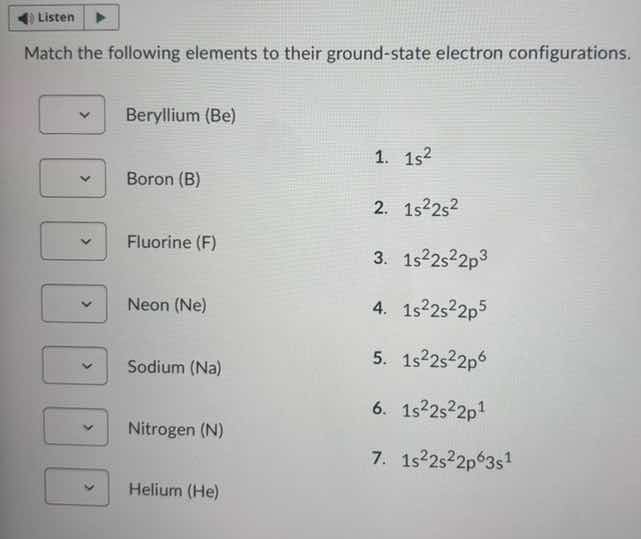
match the following elements to their ground - state electron configurations. beryllium (be) 1. $1s^2$ boron (b) 2. $1s^22s^2$ fluorine (f) 3. $1s^22s^22p^3$ neon (ne) 4. $1s^22s^22p^5$ sodium (na) 5. $1s^22s^22p^6$ nitrogen (n) 6. $1s^22s^22p^1$ helium (he) 7. $1s^22s^22p^63s^1$
Brief Explanations
Match each element to its ground-state electron configuration by using the atomic number (number of electrons) of each element and filling orbitals in order (1s, 2s, 2p, 3s).
- Beryllium (Be) has 4 electrons: fills 1s² then 2s².
- Boron (B) has 5 electrons: fills 1s²2s² then 1 electron in 2p.
- Fluorine (F) has 9 electrons: fills 1s²2s² then 5 electrons in 2p.
- Neon (Ne) has 10 electrons: fills 1s²2s² then all 6 2p orbitals.
- Sodium (Na) has 11 electrons: fills 1s²2s²2p⁶ then 1 electron in 3s.
- Nitrogen (N) has 7 electrons: fills 1s²2s² then 3 electrons in 2p.
- Helium (He) has 2 electrons: fills only the 1s orbital.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Beryllium (Be): 2. $1s^22s^2$
- Boron (B): 6. $1s^22s^22p^1$
- Fluorine (F): 4. $1s^22s^22p^5$
- Neon (Ne): 5. $1s^22s^22p^6$
- Sodium (Na): 7. $1s^22s^22p^63s^1$
- Nitrogen (N): 3. $1s^22s^22p^3$
- Helium (He): 1. $1s^2$