QUESTION IMAGE
Question
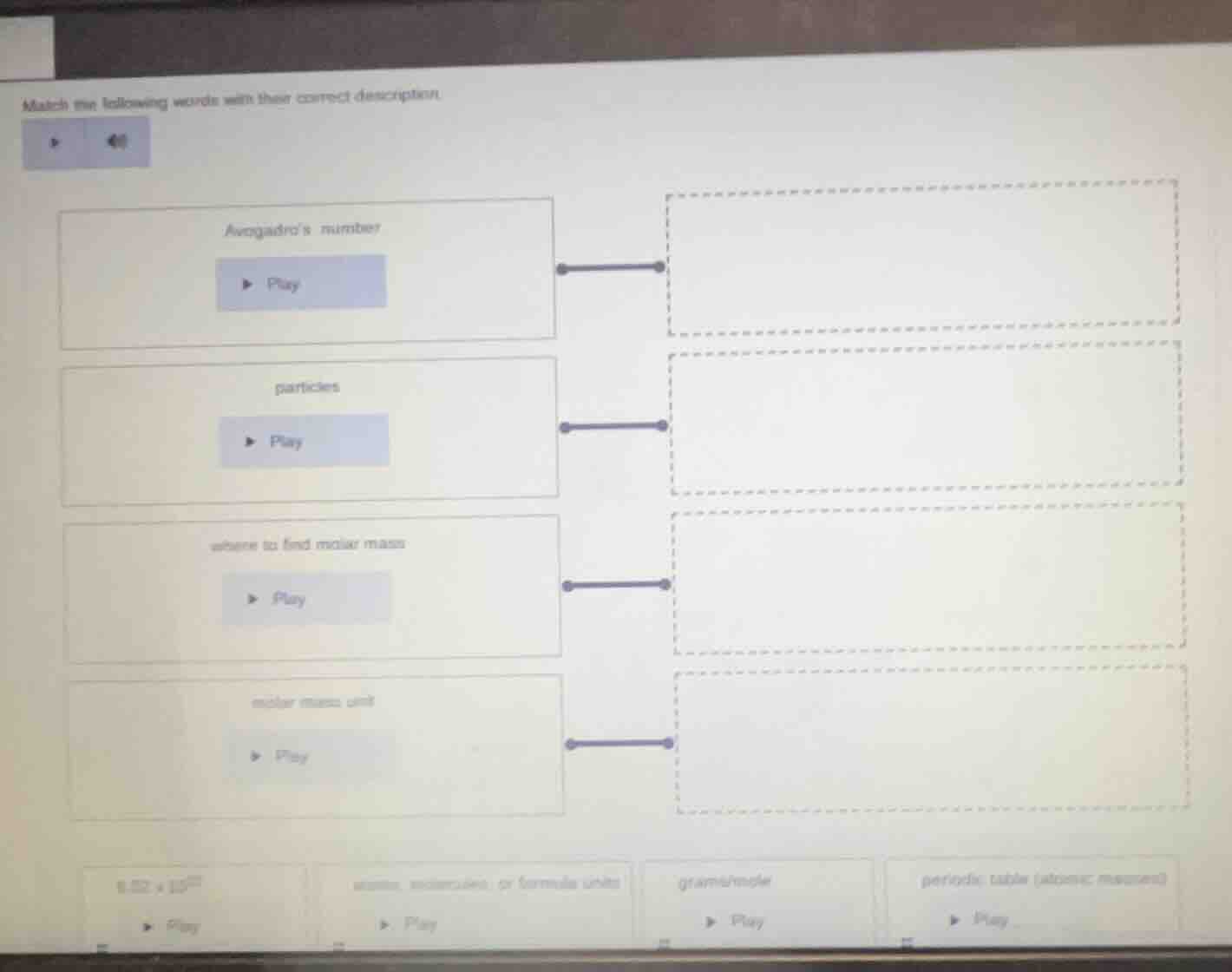
match the following words with their correct description
avogadros number
play
particles
play
where to find molar mass
play
molar mass unit
play
6.02×10²³
play
atoms, molecules, or formula units
play
gram/mole
play
periodic table (atomic mass)
play
Brief Explanations
To solve this matching problem, we analyze each term:
- Avogadro’s number is defined as \( 6.02 \times 10^{23} \), so it matches with \( 6.02 \times 10^{23} \).
- Particles in the context of moles refer to atoms, molecules, or formula units (the basic entities counted by moles), so "particles" matches with "atoms, molecules, or formula units".
- Molar mass is found from the periodic table (using atomic masses), so "where to find molar mass" matches with "periodic table (atomic masses)".
- The unit of molar mass is grams per mole (g/mol), so "molar mass unit" matches with "gram/mole".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Avogadro’s number: \( 6.02 \times 10^{23} \)
- particles: atoms, molecules, or formula units
- where to find molar mass: periodic table (atomic masses)
- molar mass unit: gram/mole