QUESTION IMAGE
Question
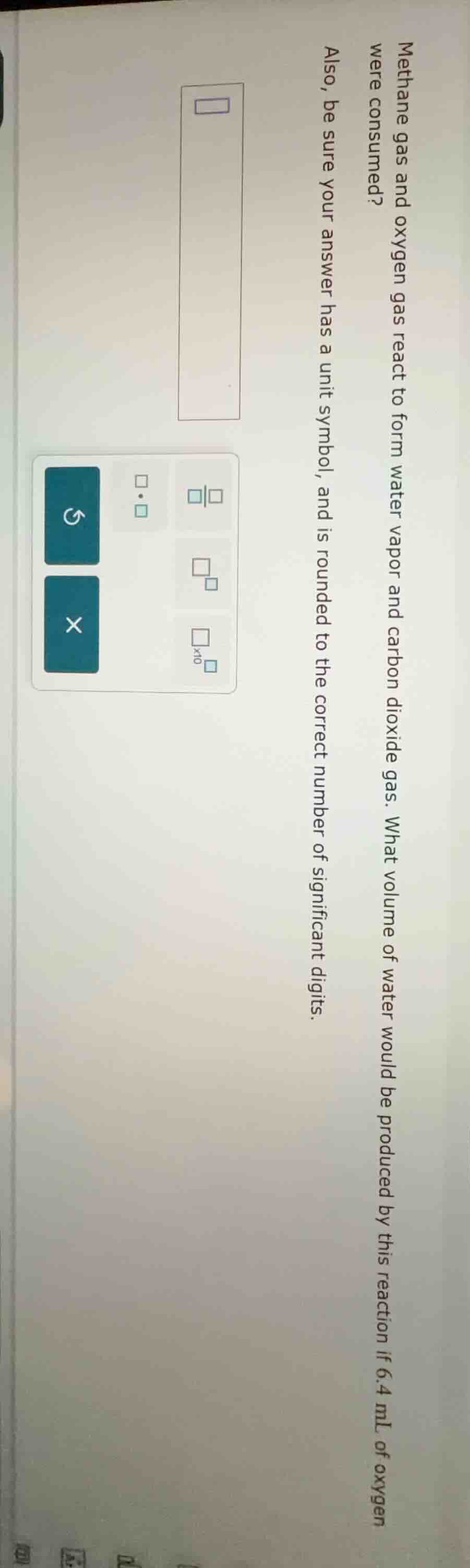
methane gas and oxygen gas react to form water vapor and carbon dioxide gas. what volume of water would be produced by this reaction if 6.4 ml of oxygen were consumed? also, be sure your answer has a unit symbol, and is rounded to the correct number of significant digits.
Step1: Write the balanced reaction
The reaction between methane ($CH_4$) and oxygen ($O_2$) is a combustion reaction. The balanced chemical equation is:
From the balanced equation, we can see the mole ratio (and for gases, volume ratio at the same conditions) of $O_2$ to $H_2O$ is $2:2$ or simplified $1:1$.
Step2: Determine the volume of water vapor
Given the volume of $O_2$ is $6.4$ mL. From the stoichiometry of the reaction (volume ratio of $O_2$ to $H_2O$ is $2:2$ or $1:1$ when considering the coefficients), the volume of $H_2O$ (water vapor) produced will be related to the volume of $O_2$ consumed.
From the balanced equation, 2 moles (or volume) of $O_2$ produce 2 moles (or volume) of $H_2O$. So the volume ratio of $O_2$ to $H_2O$ is $2:2 = 1:1$. Wait, no, let's check again. The coefficient of $O_2$ is 2 and coefficient of $H_2O$ is 2. So the ratio of volume of $O_2$ to $H_2O$ is $2:2$ or $1:1$. Wait, actually, the balanced equation is $CH_4 + 2O_2
ightarrow CO_2 + 2H_2O$. So for every 2 volumes of $O_2$ consumed, 2 volumes of $H_2O$ are produced. So the volume of $H_2O$ is equal to the volume of $O_2$ consumed? Wait, no, let's do it properly.
Let the volume of $H_2O$ be $V$.
From the balanced equation:
$$\frac{\text{Volume of } O_2}{\text{Volume of } H_2O} = \frac{2}{2} = 1$$
So $V = 6.4$ mL? Wait, no, wait the ratio of $O_2$ to $H_2O$ is $2:2$, so if we have 6.4 mL of $O_2$, then:
$$\frac{2}{2}=\frac{6.4\ mL}{V_{H_2O}}$$
Cross - multiplying, we get $2\times V_{H_2O}=2\times6.4\ mL$
So $V_{H_2O} = 6.4\ mL$? Wait, no, that seems off. Wait, maybe I made a mistake. Wait the balanced equation is $CH_4 + 2O_2
ightarrow CO_2 + 2H_2O$. So the mole ratio of $O_2$ to $H_2O$ is $2:2$ or $1:1$. So the volume of $H_2O$ produced is equal to the volume of $O_2$ consumed? Wait, if 2 L of $O_2$ produces 2 L of $H_2O$, then 6.4 mL of $O_2$ will produce 6.4 mL of $H_2O$? Wait, no, that can't be. Wait, maybe the ratio is 2 (O₂) : 2 (H₂O), so the volume of H₂O is equal to the volume of O₂. Wait, let's check with moles. At the same temperature and pressure, volume is proportional to moles (Avogadro's law). So moles of O₂ = volume of O₂ / molar volume, moles of H₂O = volume of H₂O / molar volume. From the balanced equation, moles of O₂ / moles of H₂O = 2/2 = 1. So (volume of O₂ / molar volume) / (volume of H₂O / molar volume) = 1. So volume of O₂ / volume of H₂O = 1. So volume of H₂O = volume of O₂. Wait, but that would mean 6.4 mL of O₂ produces 6.4 mL of H₂O. But let's check the coefficients again. The coefficient of O₂ is 2, coefficient of H₂O is 2. So the ratio is 2:2, so 1:1. So yes, the volume of H₂O is equal to the volume of O₂ consumed. Wait, but maybe the question has a typo or I misread. Wait, the problem says "if 6.4 mL of oxygen were consumed". So according to the stoichiometry, the volume of water vapor produced is 6.4 mL? Wait, no, wait the ratio of O₂ to H₂O is 2:2, so for every 2 parts of O₂, 2 parts of H₂O. So if we have 6.4 mL of O₂, the volume of H₂O is (2/2)*6.4 mL = 6.4 mL? Wait, that seems correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boxed{6.4\ mL}$