QUESTION IMAGE
Question
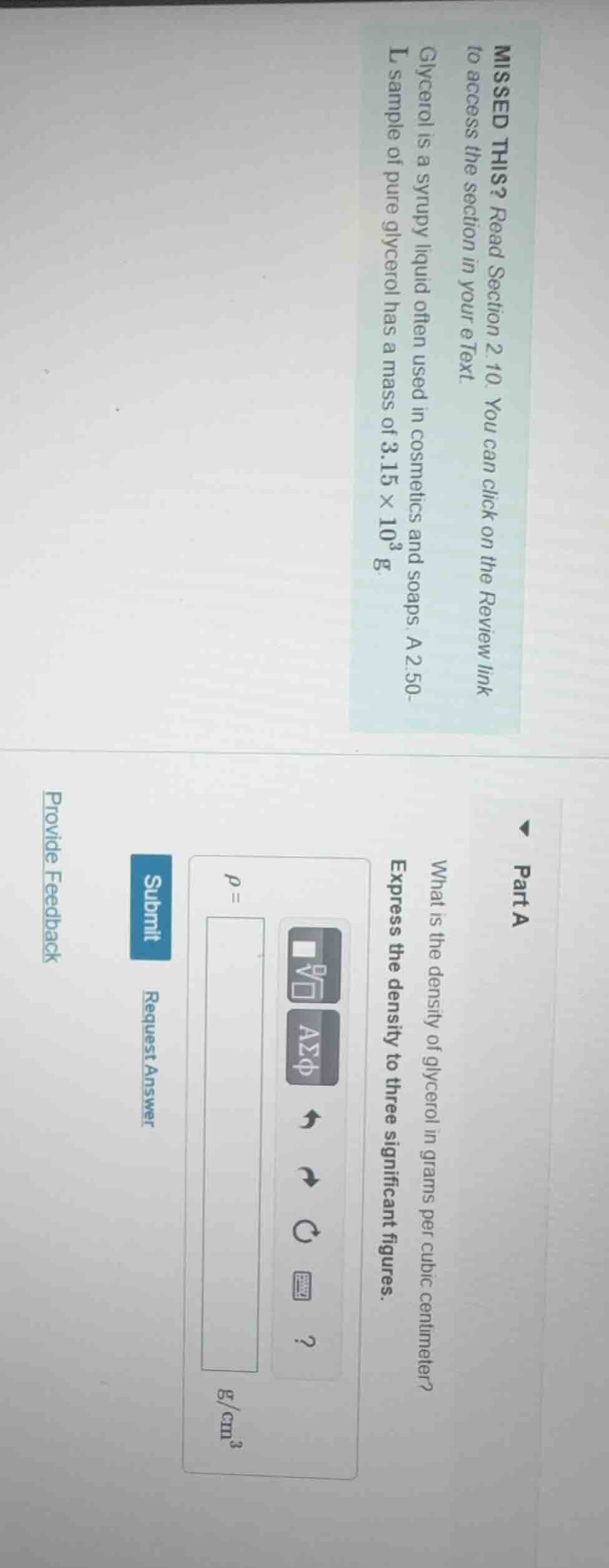
missed this? read section 2.10. you can click on the review link to access the section in your etext. glycerol is a syrupy liquid often used in cosmetics and soaps. a 2.50 - l sample of pure glycerol has a mass of 3.15×10³ g. part a what is the density of glycerol in grams per cubic centimeter? express the density to three significant figures. \\(\
ho = \\) \\(\frac{g}{cm^3}\\) submit request answer provide feedback
Step1: Convert volume to cm³
Since \(1\ \text{L} = 1000\ \text{cm}^3\),
\(2.50\ \text{L} = 2.50 \times 1000 = 2500\ \text{cm}^3\)
Step2: Apply density formula
Density \(
ho = \frac{\text{mass}}{\text{volume}}\)
\(
ho = \frac{3.15 \times 10^3\ \text{g}}{2500\ \text{cm}^3}\)
Step3: Calculate and round to 3 sig figs
\(
ho = \frac{3150}{2500} = 1.26\ \text{g/cm}^3\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(1.26\ \text{g/cm}^3\)