QUESTION IMAGE
Question
mole ratio
mole conversion
given amount
moles of unknown
moles of known
molar mass (g) of unknown
1 mole of unknown
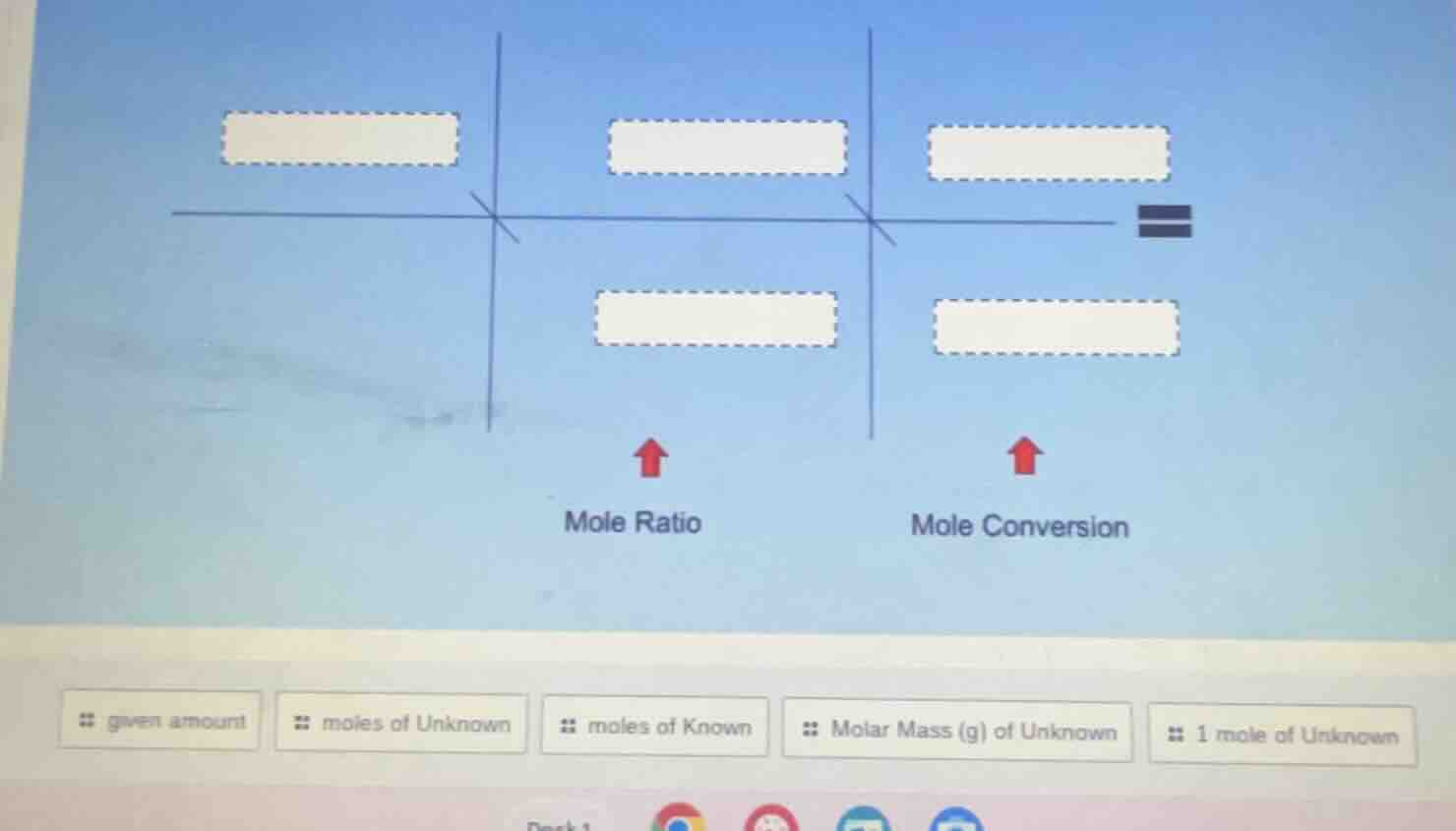
This is a stoichiometry mole conversion flow. First, start with the given amount of the known substance, convert it to moles of the known. Then use the mole ratio (moles of unknown over moles of known) to convert to moles of the unknown. Finally, use the molar mass of the unknown (or the conversion of 1 mole of unknown to mass) to get the final amount of the unknown.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Top left blank: given amount
- Top middle blank: moles of Known
- Bottom middle blank: moles of Unknown
- Top right blank: 1 mole of Unknown
- Bottom right blank: Molar Mass (g) of Unknown
The full equation structure is:
$\text{given amount} \times \frac{\text{moles of Known}}{\text{[molar mass of known, implied]}} \times \frac{\text{moles of Unknown}}{\text{moles of Known}} \times \frac{\text{Molar Mass (g) of Unknown}}{1 \text{ mole of Unknown}} = \text{mass of unknown}$