QUESTION IMAGE
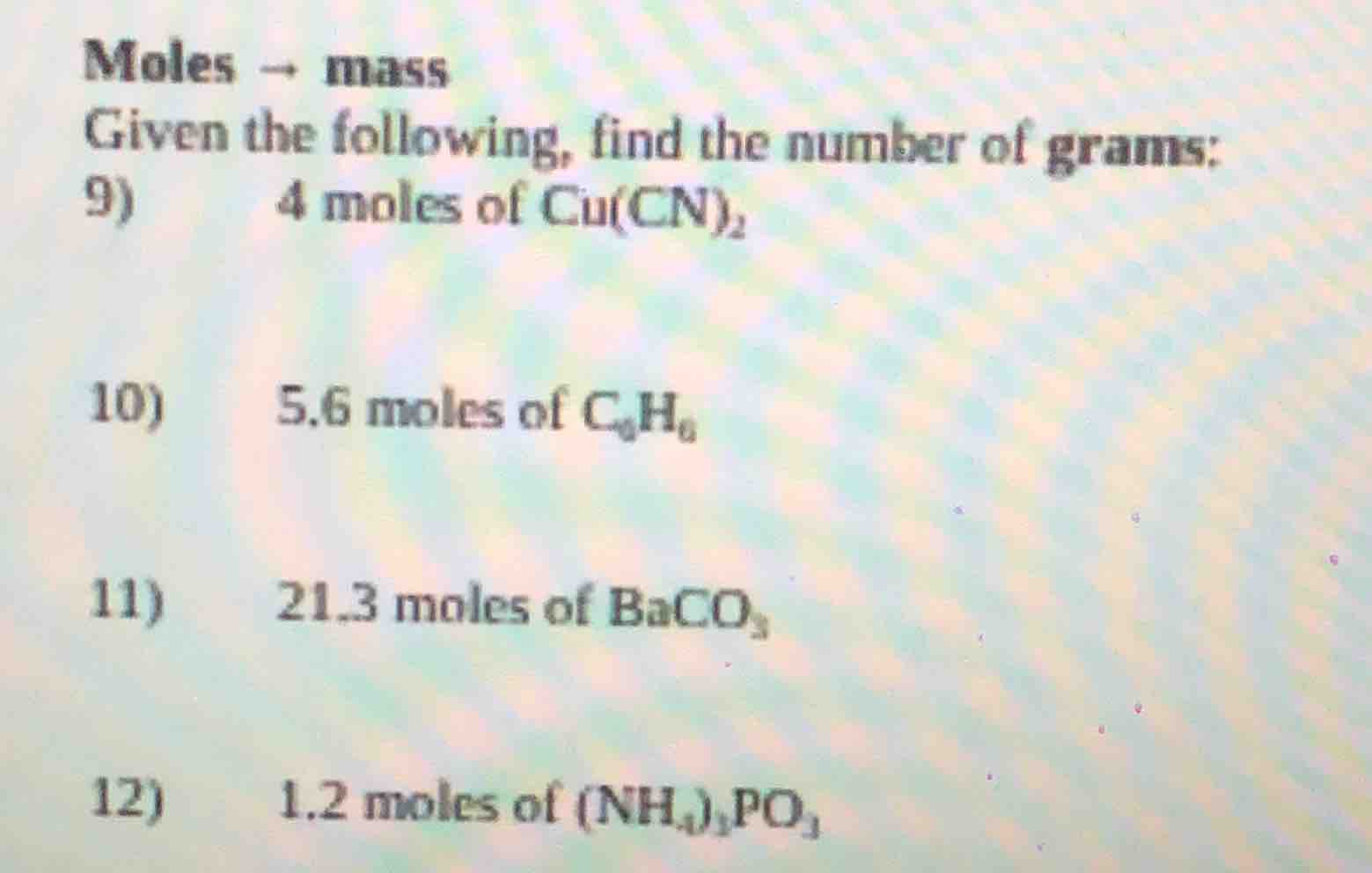
Question
moles → mass
given the following, find the number of grams:
- 4 moles of cu(cn)₂
- 5.6 moles of c₆h₆
- 21.3 moles of baco₃
- 1.2 moles of (nh₄)₂po₃
Problem 9: 4 moles of $\ce{Cu(CN)_{2}}$
Step 1: Calculate molar mass of $\ce{Cu(CN)_{2}}$
Molar mass of $\ce{Cu}$: $63.55\ \text{g/mol}$, $\ce{C}$: $12.01\ \text{g/mol}$, $\ce{N}$: $14.01\ \text{g/mol}$.
For $\ce{Cu(CN)_{2}}$: $1\times63.55 + 2\times(12.01 + 14.01) = 63.55 + 2\times26.02 = 63.55 + 52.04 = 115.59\ \text{g/mol}$.
Step 2: Use $m = n\times M$ (mass = moles × molar mass)
$n = 4\ \text{mol}$, $M = 115.59\ \text{g/mol}$.
$m = 4\times115.59 = 462.36\ \text{g}$.
Step 1: Calculate molar mass of $\ce{C_{6}H_{6}}$
Molar mass of $\ce{C}$: $12.01\ \text{g/mol}$, $\ce{H}$: $1.008\ \text{g/mol}$.
For $\ce{C_{6}H_{6}}$: $6\times12.01 + 6\times1.008 = 72.06 + 6.048 = 78.108\ \text{g/mol}$.
Step 2: Use $m = n\times M$
$n = 5.6\ \text{mol}$, $M = 78.108\ \text{g/mol}$.
$m = 5.6\times78.108 \approx 437.40\ \text{g}$.
Step 1: Calculate molar mass of $\ce{BaCO_{3}}$
Molar mass of $\ce{Ba}$: $137.33\ \text{g/mol}$, $\ce{C}$: $12.01\ \text{g/mol}$, $\ce{O}$: $16.00\ \text{g/mol}$.
For $\ce{BaCO_{3}}$: $137.33 + 12.01 + 3\times16.00 = 137.33 + 12.01 + 48.00 = 197.34\ \text{g/mol}$.
Step 2: Use $m = n\times M$
$n = 21.3\ \text{mol}$, $M = 197.34\ \text{g/mol}$.
$m = 21.3\times197.34 \approx 4203.34\ \text{g}$ (or ~4.20×10³ g).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$462.36\ \text{g}$ (or ~462 g, depending on sig figs)