QUESTION IMAGE
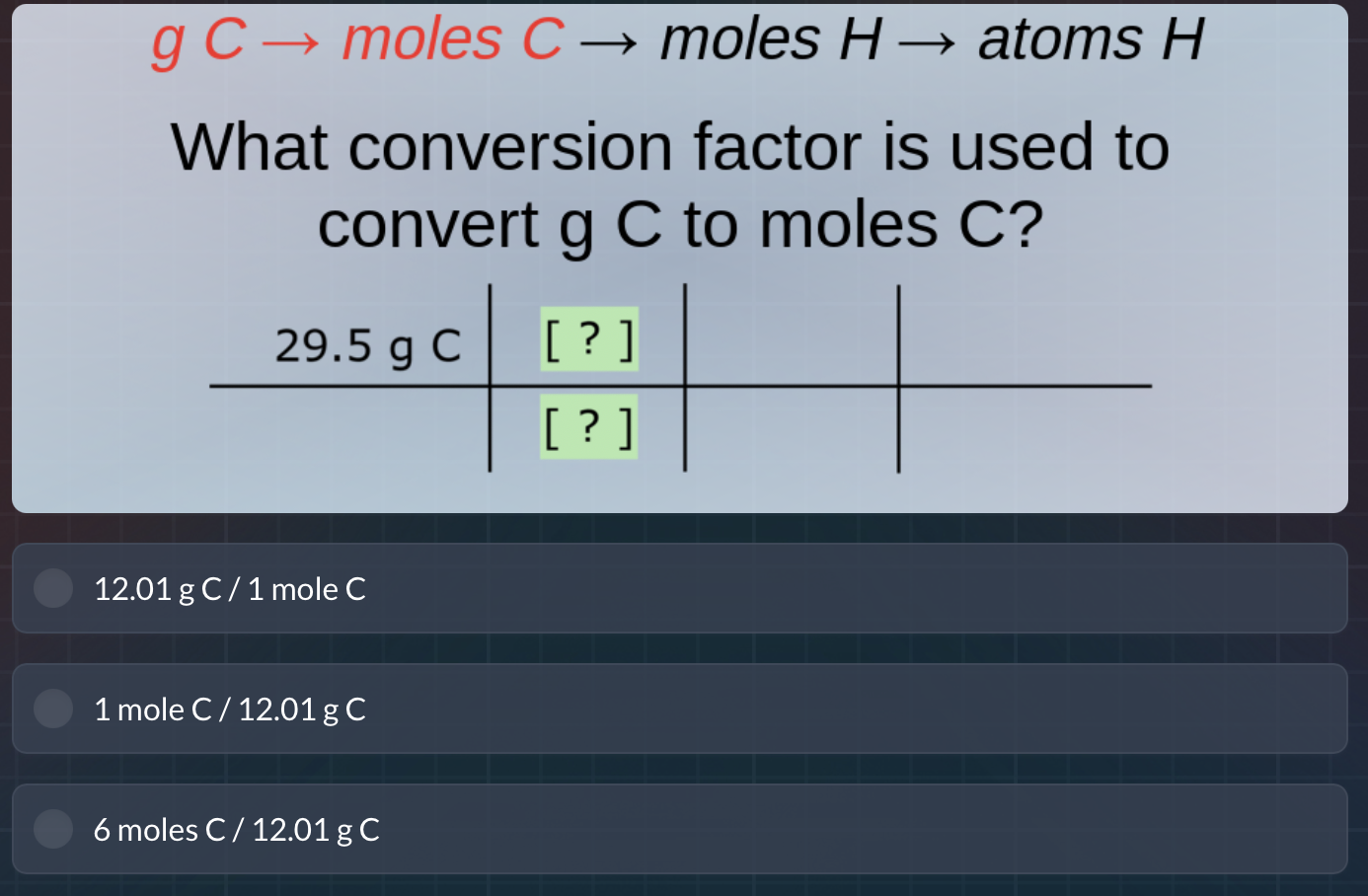
Question
g c → moles c → moles h → atoms h
what conversion factor is used to
convert g c to moles c?
29.5 g c ?
?
12.01 g c / 1 mole c
1 mole c / 12.01 g c
6 moles c / 12.01 g c
Step1: Recall molar mass conversion
To convert grams to moles, we use the molar mass (g/mol) as a conversion factor. The molar mass of carbon (C) is approximately 12.01 g/mol, meaning 1 mole of C has a mass of 12.01 g.
Step2: Determine the conversion factor direction
To convert grams (g) of C to moles (mol) of C, we need a factor where grams cancel out. So the numerator should be moles and the denominator grams. Thus, the conversion factor is $\frac{1\ \text{mole C}}{12.01\ \text{g C}}$, because when we multiply grams of C by this factor, the grams unit cancels, leaving moles of C.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1 mole C / 12.01 g C