QUESTION IMAGE
Question
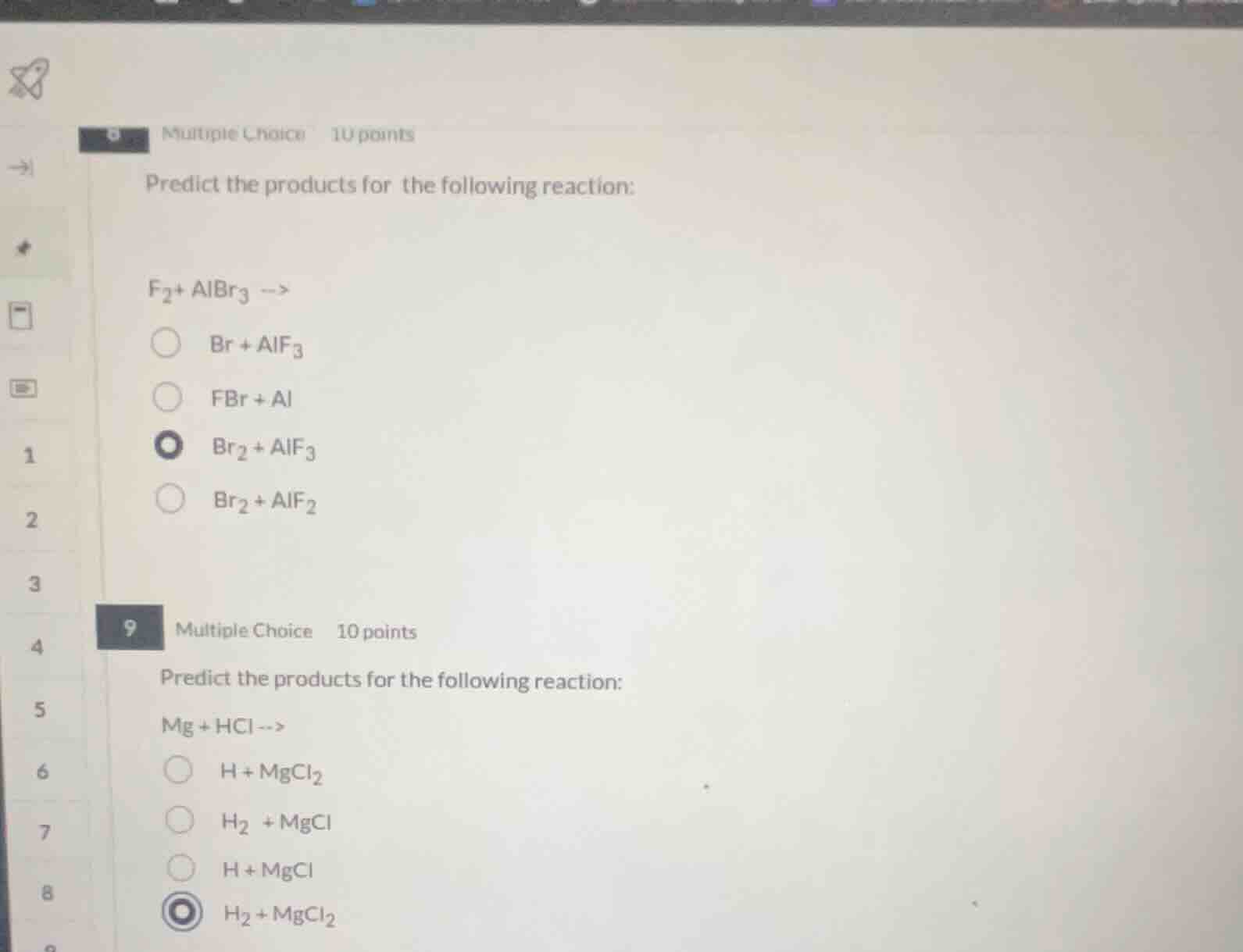
5 multiple choice 10 points predict the products for the following reaction: f₂ + albr₃ ---> br + alf₃ fbr + al br₂ + alf₃ br₂ + alf₂ 9 multiple choice 10 points predict the products for the following reaction: mg + hcl ---> h + mgcl₂ h₂ + mgcl h + mgcl h₂ + mgcl₂
Step1: Analyze first reaction (displacement)
Fluorine (F₂) is more reactive than bromine, so it displaces Br from AlBr₃. Balance the reaction:
$$3\text{F}_2 + 2\text{AlBr}_3
ightarrow 3\text{Br}_2 + 2\text{AlF}_3$$
Step2: Analyze second reaction (single displacement)
Magnesium reacts with hydrochloric acid to produce hydrogen gas and magnesium chloride. Balance the reaction:
$$\text{Mg} + 2\text{HCl}
ightarrow \text{H}_2 + \text{MgCl}_2$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{Br}_2 + \text{AlF}_3$
- $\text{H}_2 + \text{MgCl}_2$