QUESTION IMAGE
Question
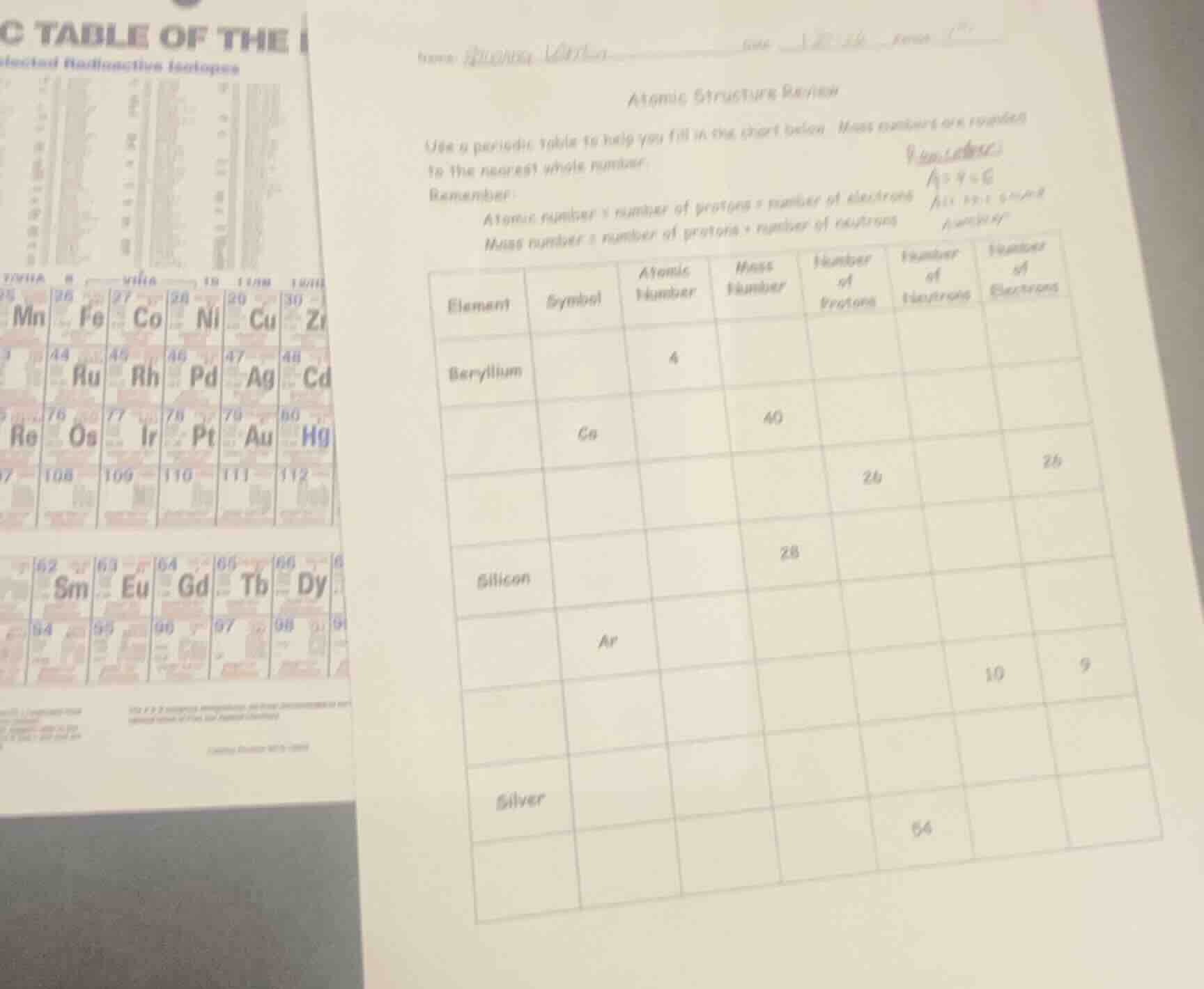
name:
atomic structure review
use a periodic table to help you fill in the chart below. mass numbers are rounded to the nearest whole number.
remember:
atomic number = number of protons = number of electrons
mass number = number of protons + number of neutrons
| element | symbol | atomic number | mass number | number of protons | number of neutrons | number of electrons |
|---|---|---|---|---|---|---|
| ga | 40 | |||||
| 26 | 26 | |||||
| silicon | 28 | |||||
| ar | ||||||
| 10 | 9 | |||||
| silver | 64 | |||||
To solve the atomic structure chart, we use the following rules:
- Atomic number = number of protons = number of electrons (in neutral atoms).
- Mass number = number of protons + number of neutrons.
1. Beryllium
- Symbol: Be (from periodic table, atomic number 4 is Beryllium).
- Atomic Number: 4 (given).
- Number of Protons: 4 (atomic number = protons).
- Number of Electrons: 4 (protons = electrons in neutral atoms).
- Mass Number: From periodic table, Beryllium’s mass number is ~9 (neutrons = mass number – protons = \( 9 - 4 = 5 \)).
2. Ga (Gallium)
- Element: Gallium (symbol Ga).
- Atomic Number: From periodic table, Ga has atomic number 31.
- Number of Protons: 31 (atomic number = protons).
- Number of Electrons: 31 (protons = electrons).
- Number of Neutrons: Mass number (40) – protons (31) = \( 40 - 31 = 9 \).
3. Element with 26 Protons/Electrons
- Element: Iron (Fe, atomic number 26).
- Symbol: Fe.
- Atomic Number: 26 (protons = 26).
- Mass Number: From periodic table, Fe’s mass number is ~56 (neutrons = \( 56 - 26 = 30 \)).
4. Silicon (Mass Number 28)
- Symbol: Si (Silicon).
- Atomic Number: 14 (Si has atomic number 14).
- Number of Protons: 14 (atomic number = protons).
- Number of Electrons: 14 (protons = electrons).
- Number of Neutrons: Mass number (28) – protons (14) = \( 28 - 14 = 14 \).
5. Ar (Argon)
- Element: Argon (symbol Ar).
- Atomic Number: 18 (Ar has atomic number 18).
- Number of Protons: 18 (atomic number = protons).
- Number of Electrons: 18 (protons = electrons).
- Mass Number: From periodic table, Ar’s mass number is ~40 (neutrons = \( 40 - 18 = 22 \)).
6. Element with 10 Neutrons, 9 Electrons
- Atomic Number: 9 (electrons = 9, so protons = 9).
- Element: Fluorine (F, atomic number 9).
- Symbol: F.
- Mass Number: Protons (9) + neutrons (10) = \( 9 + 10 = 19 \).
7. Silver
- Symbol: Ag (Silver).
- Atomic Number: 47 (Ag has atomic number 47).
- Number of Protons: 47 (atomic number = protons).
- Number of Electrons: 47 (protons = electrons).
- Mass Number: Protons (47) + neutrons (54) = \( 47 + 54 = 101 \).
Filled Chart (Summary):
| Element | Symbol | Atomic Number | Mass Number | Protons | Neutrons | Electrons |
|---|---|---|---|---|---|---|
| Gallium | Ga | 31 | 40 | 31 | 9 | 31 |
| Iron | Fe | 26 | 56 | 26 | 30 | 26 |
| Silicon | Si | 14 | 28 | 14 | 14 | 14 |
| Argon | Ar | 18 | 40 | 18 | 22 | 18 |
| Fluorine | F | 9 | 19 | 9 | 10 | 9 |
| Silver | Ag | 47 | 101 | 47 | 54 | 47 |
(Note: Mass numbers are approximate from the periodic table, rounded to whole numbers.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve the atomic structure chart, we use the following rules:
- Atomic number = number of protons = number of electrons (in neutral atoms).
- Mass number = number of protons + number of neutrons.
1. Beryllium
- Symbol: Be (from periodic table, atomic number 4 is Beryllium).
- Atomic Number: 4 (given).
- Number of Protons: 4 (atomic number = protons).
- Number of Electrons: 4 (protons = electrons in neutral atoms).
- Mass Number: From periodic table, Beryllium’s mass number is ~9 (neutrons = mass number – protons = \( 9 - 4 = 5 \)).
2. Ga (Gallium)
- Element: Gallium (symbol Ga).
- Atomic Number: From periodic table, Ga has atomic number 31.
- Number of Protons: 31 (atomic number = protons).
- Number of Electrons: 31 (protons = electrons).
- Number of Neutrons: Mass number (40) – protons (31) = \( 40 - 31 = 9 \).
3. Element with 26 Protons/Electrons
- Element: Iron (Fe, atomic number 26).
- Symbol: Fe.
- Atomic Number: 26 (protons = 26).
- Mass Number: From periodic table, Fe’s mass number is ~56 (neutrons = \( 56 - 26 = 30 \)).
4. Silicon (Mass Number 28)
- Symbol: Si (Silicon).
- Atomic Number: 14 (Si has atomic number 14).
- Number of Protons: 14 (atomic number = protons).
- Number of Electrons: 14 (protons = electrons).
- Number of Neutrons: Mass number (28) – protons (14) = \( 28 - 14 = 14 \).
5. Ar (Argon)
- Element: Argon (symbol Ar).
- Atomic Number: 18 (Ar has atomic number 18).
- Number of Protons: 18 (atomic number = protons).
- Number of Electrons: 18 (protons = electrons).
- Mass Number: From periodic table, Ar’s mass number is ~40 (neutrons = \( 40 - 18 = 22 \)).
6. Element with 10 Neutrons, 9 Electrons
- Atomic Number: 9 (electrons = 9, so protons = 9).
- Element: Fluorine (F, atomic number 9).
- Symbol: F.
- Mass Number: Protons (9) + neutrons (10) = \( 9 + 10 = 19 \).
7. Silver
- Symbol: Ag (Silver).
- Atomic Number: 47 (Ag has atomic number 47).
- Number of Protons: 47 (atomic number = protons).
- Number of Electrons: 47 (protons = electrons).
- Mass Number: Protons (47) + neutrons (54) = \( 47 + 54 = 101 \).
Filled Chart (Summary):
| Element | Symbol | Atomic Number | Mass Number | Protons | Neutrons | Electrons |
|---|---|---|---|---|---|---|
| Gallium | Ga | 31 | 40 | 31 | 9 | 31 |
| Iron | Fe | 26 | 56 | 26 | 30 | 26 |
| Silicon | Si | 14 | 28 | 14 | 14 | 14 |
| Argon | Ar | 18 | 40 | 18 | 22 | 18 |
| Fluorine | F | 9 | 19 | 9 | 10 | 9 |
| Silver | Ag | 47 | 101 | 47 | 54 | 47 |
(Note: Mass numbers are approximate from the periodic table, rounded to whole numbers.)