QUESTION IMAGE
Question
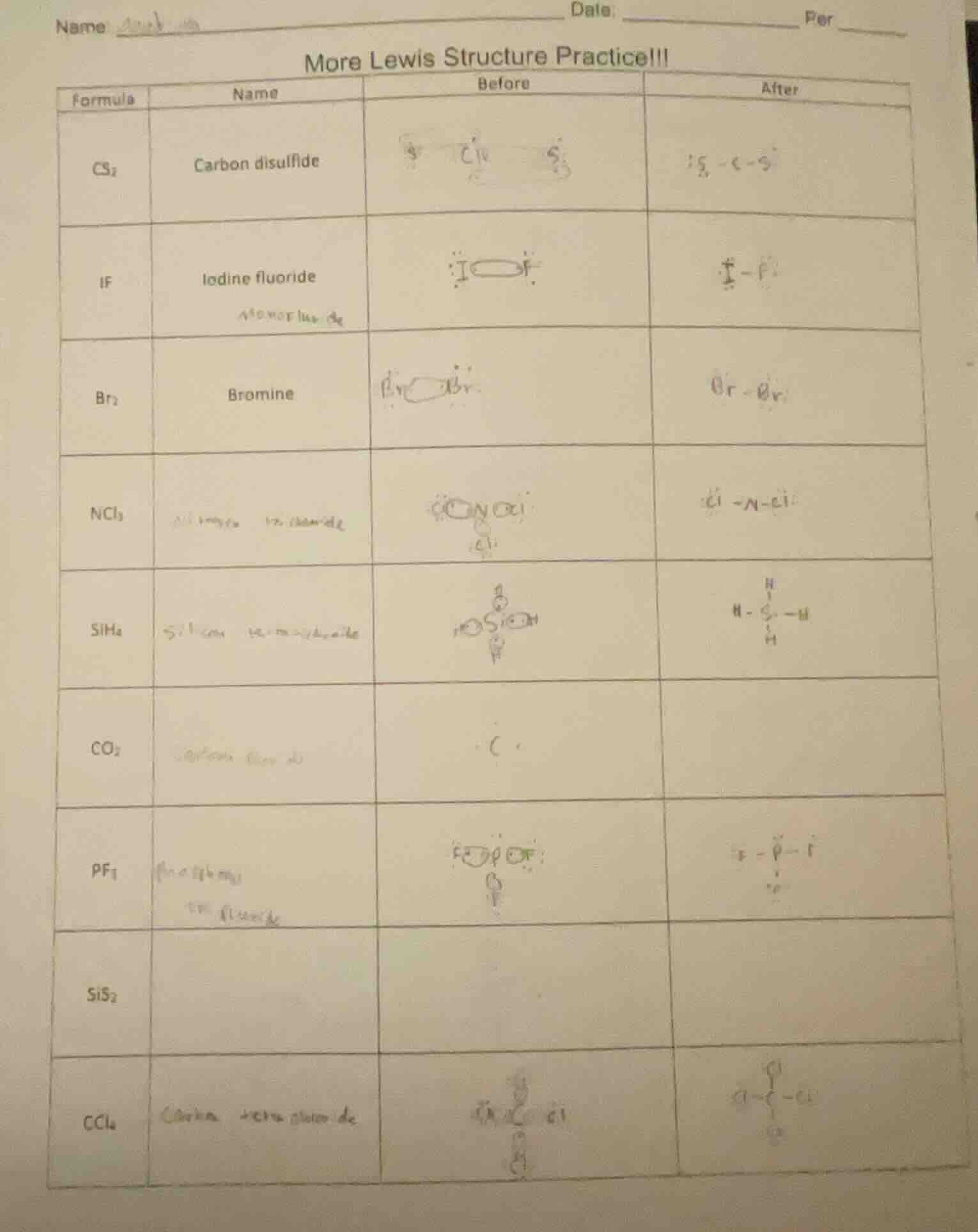
name date: per more lewis structure practice!!! formula name before after cs₂ carbon disulfide if iodine fluoride br₂ bromine ncl₃ sih₄ co₂ pf₃ sis₂ ccl₄
To determine the correct Lewis structures or complete the table, we analyze each compound:
1. \( \boldsymbol{CS_2} \) (Carbon disulfide)
- Valence electrons: \( C \) (4) + \( 2 \times S \) (6 each) = \( 4 + 12 = 16 \).
- Structure: \( C \) is central, double - bonded to two \( S \) atoms (\( S = C = S \)), with 2 lone pairs on each \( S \).
2. \( \boldsymbol{IF} \) (Iodine fluoride)
- Valence electrons: \( I \) (7) + \( F \) (7) = \( 14 \).
- Structure: Single bond between \( I \) and \( F \), with 3 lone pairs on each (\( I - F \), each has 3 lone pairs).
3. \( \boldsymbol{Br_2} \) (Bromine)
- Valence electrons: \( 2 \times Br \) (7 each) = \( 14 \).
- Structure: Single bond between two \( Br \) atoms (\( Br - Br \)), with 3 lone pairs on each.
4. \( \boldsymbol{NCl_3} \) (Nitrogen trichloride)
- Valence electrons: \( N \) (5) + \( 3 \times Cl \) (7 each) = \( 5+21 = 26 \).
- Structure: \( N \) is central, single - bonded to 3 \( Cl \) atoms, with 1 lone pair on \( N \), 3 lone pairs on each \( Cl \) (\( \overset{..}{N}-Cl \), \( Cl \) has 3 lone pairs).
5. \( \boldsymbol{SiH_4} \) (Silane)
- Valence electrons: \( Si \) (4) + \( 4 \times H \) (1 each) = \( 8 \).
- Structure: \( Si \) is central, single - bonded to 4 \( H \) atoms (tetrahedral, \( H - Si - H \) with \( H \) above and below \( Si \)).
6. \( \boldsymbol{CO_2} \) (Carbon dioxide)
- Valence electrons: \( C \) (4) + \( 2 \times O \) (6 each) = \( 16 \).
- Structure: \( C \) is central, double - bonded to two \( O \) atoms (\( O = C = O \)), with 2 lone pairs on each \( O \).
7. \( \boldsymbol{PF_3} \) (Phosphorus trifluoride)
- Valence electrons: \( P \) (5) + \( 3 \times F \) (7 each) = \( 5 + 21=26 \).
- Structure: \( P \) is central, single - bonded to 3 \( F \) atoms, with 1 lone pair on \( P \), 3 lone pairs on each \( F \) (\( F - P - F \), \( F \) has 3 lone pairs, \( P \) has 1 lone pair).
8. \( \boldsymbol{SiS_2} \) (Silicon disulfide)
- Valence electrons: \( Si \) (4) + \( 2 \times S \) (6 each) = \( 4+12 = 16 \).
- Structure: \( Si \) is central, double - bonded to two \( S \) atoms (\( S = Si = S \)), with 2 lone pairs on each \( S \).
9. \( \boldsymbol{CCl_4} \) (Carbon tetrachloride)
- Valence electrons: \( C \) (4) + \( 4 \times Cl \) (7 each) = \( 4 + 28=32 \).
- Structure: \( C \) is central, single - bonded to 4 \( Cl \) atoms (tetrahedral), with 3 lone pairs on each \( Cl \).
For the "Name" column:
- \( SiH_4 \): Silane
- \( CO_2 \): Carbon dioxide
- \( PF_3 \): Phosphorus trifluoride
- \( SiS_2 \): Silicon disulfide
- \( CCl_4 \): Carbon tetrachloride
To fill the table properly, use the above - derived Lewis structures and correct names. For example, in the "After" column, draw the structures as described, and in the "Name" column, write the correct compound names.
If you want to correct a specific row (e.g., \( SiH_4 \) name is "Silane", \( CO_2 \) is "Carbon dioxide", etc.), the key is to apply valence electron counting and bonding rules (octet rule, except for \( H \) which follows duet rule) to determine the Lewis structures.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the correct Lewis structures or complete the table, we analyze each compound:
1. \( \boldsymbol{CS_2} \) (Carbon disulfide)
- Valence electrons: \( C \) (4) + \( 2 \times S \) (6 each) = \( 4 + 12 = 16 \).
- Structure: \( C \) is central, double - bonded to two \( S \) atoms (\( S = C = S \)), with 2 lone pairs on each \( S \).
2. \( \boldsymbol{IF} \) (Iodine fluoride)
- Valence electrons: \( I \) (7) + \( F \) (7) = \( 14 \).
- Structure: Single bond between \( I \) and \( F \), with 3 lone pairs on each (\( I - F \), each has 3 lone pairs).
3. \( \boldsymbol{Br_2} \) (Bromine)
- Valence electrons: \( 2 \times Br \) (7 each) = \( 14 \).
- Structure: Single bond between two \( Br \) atoms (\( Br - Br \)), with 3 lone pairs on each.
4. \( \boldsymbol{NCl_3} \) (Nitrogen trichloride)
- Valence electrons: \( N \) (5) + \( 3 \times Cl \) (7 each) = \( 5+21 = 26 \).
- Structure: \( N \) is central, single - bonded to 3 \( Cl \) atoms, with 1 lone pair on \( N \), 3 lone pairs on each \( Cl \) (\( \overset{..}{N}-Cl \), \( Cl \) has 3 lone pairs).
5. \( \boldsymbol{SiH_4} \) (Silane)
- Valence electrons: \( Si \) (4) + \( 4 \times H \) (1 each) = \( 8 \).
- Structure: \( Si \) is central, single - bonded to 4 \( H \) atoms (tetrahedral, \( H - Si - H \) with \( H \) above and below \( Si \)).
6. \( \boldsymbol{CO_2} \) (Carbon dioxide)
- Valence electrons: \( C \) (4) + \( 2 \times O \) (6 each) = \( 16 \).
- Structure: \( C \) is central, double - bonded to two \( O \) atoms (\( O = C = O \)), with 2 lone pairs on each \( O \).
7. \( \boldsymbol{PF_3} \) (Phosphorus trifluoride)
- Valence electrons: \( P \) (5) + \( 3 \times F \) (7 each) = \( 5 + 21=26 \).
- Structure: \( P \) is central, single - bonded to 3 \( F \) atoms, with 1 lone pair on \( P \), 3 lone pairs on each \( F \) (\( F - P - F \), \( F \) has 3 lone pairs, \( P \) has 1 lone pair).
8. \( \boldsymbol{SiS_2} \) (Silicon disulfide)
- Valence electrons: \( Si \) (4) + \( 2 \times S \) (6 each) = \( 4+12 = 16 \).
- Structure: \( Si \) is central, double - bonded to two \( S \) atoms (\( S = Si = S \)), with 2 lone pairs on each \( S \).
9. \( \boldsymbol{CCl_4} \) (Carbon tetrachloride)
- Valence electrons: \( C \) (4) + \( 4 \times Cl \) (7 each) = \( 4 + 28=32 \).
- Structure: \( C \) is central, single - bonded to 4 \( Cl \) atoms (tetrahedral), with 3 lone pairs on each \( Cl \).
For the "Name" column:
- \( SiH_4 \): Silane
- \( CO_2 \): Carbon dioxide
- \( PF_3 \): Phosphorus trifluoride
- \( SiS_2 \): Silicon disulfide
- \( CCl_4 \): Carbon tetrachloride
To fill the table properly, use the above - derived Lewis structures and correct names. For example, in the "After" column, draw the structures as described, and in the "Name" column, write the correct compound names.
If you want to correct a specific row (e.g., \( SiH_4 \) name is "Silane", \( CO_2 \) is "Carbon dioxide", etc.), the key is to apply valence electron counting and bonding rules (octet rule, except for \( H \) which follows duet rule) to determine the Lewis structures.