QUESTION IMAGE
Question
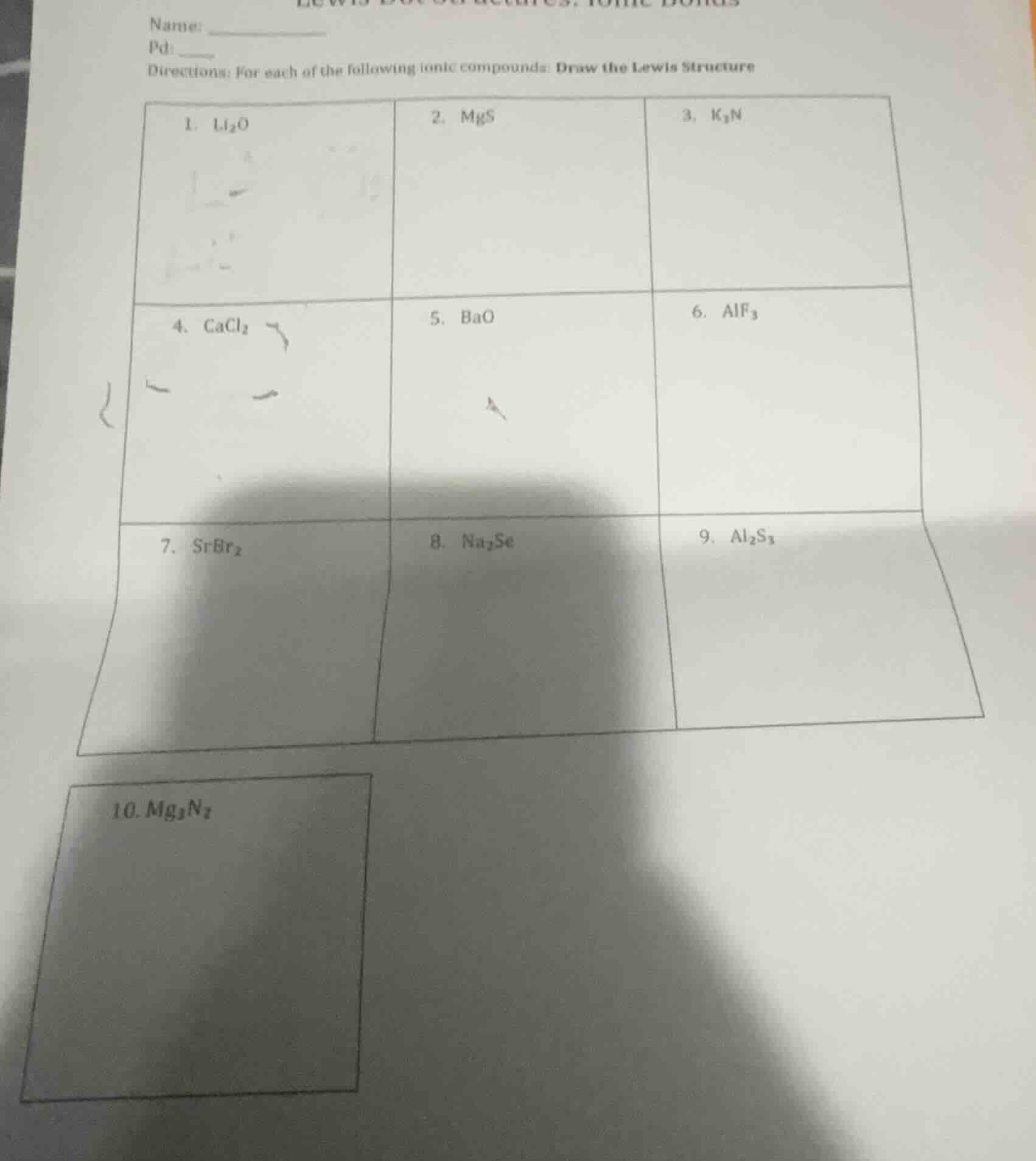
name: ______
pd: ____
directions: for each of the following ionic compounds: draw the lewis structure
- li₂o\t2. mgs\t3. k₃n
- cacl₂\t5. bao\t6. alf₃
- srbr₂\t8. na₂se\t9. al₂s₃
- mg₃n₂
To draw the Lewis structures for these ionic compounds, we follow these general steps for ionic compounds (composed of cations and anions):
Step 1: Identify the ions
For an ionic compound \( M_xA_y \), the cation is \( M^{n+} \) and the anion is \( A^{m-} \), where \( n \) and \( m \) are the charges (determined by valence electrons or group number).
Step 2: Determine charges
- Group 1 metals (Li, Na, K, etc.): \( +1 \) charge.
- Group 2 metals (Mg, Ca, Ba, Sr, etc.): \( +2 \) charge.
- Group 13 metals (Al): \( +3 \) charge.
- Group 15 nonmetals (N, P, etc.): \( -3 \) charge (gain 3 electrons to fill octet).
- Group 16 nonmetals (O, S, Se, etc.): \( -2 \) charge (gain 2 electrons).
- Group 17 nonmetals (F, Cl, Br, etc.): \( -1 \) charge (gain 1 electron).
Example: \( \boldsymbol{\ce{Li2O}} \) (1)
- Li (Group 1): \( \ce{Li^+} \) (loses 1 electron).
- O (Group 16): \( \ce{O^{2-}} \) (gains 2 electrons).
- Charge balance: \( 2 \times (+1) + 1 \times (-2) = 0 \), so 2 \( \ce{Li^+} \) and 1 \( \ce{O^{2-}} \).
- Lewis structure: \( [\ce{Li^+}]_2 \: \: \ce{O^{2-}} \) (or draw each ion with valence electrons: Li has 0 valence electrons (lost 1), O has 8 (gained 2), so \( \ce{Li^+} \) (no dots) and \( \ce{O^{2-}} \) (8 dots, 2- charge)).
Example: \( \boldsymbol{\ce{MgS}} \) (2)
- Mg (Group 2): \( \ce{Mg^{2+}} \) (loses 2 electrons).
- S (Group 16): \( \ce{S^{2-}} \) (gains 2 electrons).
- Charge balance: \( 1 \times (+2) + 1 \times (-2) = 0 \), so 1 \( \ce{Mg^{2+}} \) and 1 \( \ce{S^{2-}} \).
- Lewis structure: \( \ce{Mg^{2+}} \: \: \ce{S^{2-}} \) (Mg has 0 valence electrons, S has 8 dots, 2- charge).
Example: \( \boldsymbol{\ce{K3N}} \) (3)
- K (Group 1): \( \ce{K^+} \) (loses 1 electron).
- N (Group 15): \( \ce{N^{3-}} \) (gains 3 electrons).
- Charge balance: \( 3 \times (+1) + 1 \times (-3) = 0 \), so 3 \( \ce{K^+} \) and 1 \( \ce{N^{3-}} \).
- Lewis structure: \( [\ce{K^+}]_3 \: \: \ce{N^{3-}} \) (K has 0 valence electrons, N has 8 dots, 3- charge).
Example: \( \boldsymbol{\ce{CaCl2}} \) (4)
- Ca (Group 2): \( \ce{Ca^{2+}} \) (loses 2 electrons).
- Cl (Group 17): \( \ce{Cl^-} \) (gains 1 electron).
- Charge balance: \( 1 \times (+2) + 2 \times (-1) = 0 \), so 1 \( \ce{Ca^{2+}} \) and 2 \( \ce{Cl^-} \).
- Lewis structure: \( \ce{Ca^{2+}} \: \: [\ce{Cl^-}]_2 \) (Ca has 0 valence electrons, each Cl has 8 dots, 1- charge).
Example: \( \boldsymbol{\ce{AlF3}} \) (6)
- Al (Group 13): \( \ce{Al^{3+}} \) (loses 3 electrons).
- F (Group 17): \( \ce{F^-} \) (gains 1 electron).
- Charge balance: \( 1 \times (+3) + 3 \times (-1) = 0 \), so 1 \( \ce{Al^{3+}} \) and 3 \( \ce{F^-} \).
- Lewis structure: \( \ce{Al^{3+}} \: \: [\ce{F^-}]_3 \) (Al has 0 valence electrons, each F has 8 dots, 1- charge).
Example: \( \boldsymbol{\ce{Al2S3}} \) (9)
- Al (Group 13): \( \ce{Al^{3+}} \) (loses 3 electrons).
- S (Group 16): \( \ce{S^{2-}} \) (gains 2 electrons).
- Charge balance: \( 2 \times (+3) + 3 \times (-2) = 0 \), so 2 \( \ce{Al^{3+}} \) and 3 \( \ce{S^{2-}} \).
- Lewis structure: \( [\ce{Al^{3+}}]_2 \: \: [\ce{S^{2-}}]_3 \) (Al has 0 valence electrons, each S has 8 dots, 2- charge).
Example: \( \boldsymbol{\ce{Mg3N2}} \) (10)
- Mg (Group 2): \( \ce{Mg^{2+}} \) (loses 2 electrons).
- N (Group 15): \( \ce{N^{3-}} \) (gains 3 electrons).
- Charge balance: \( 3 \times (+2) + 2 \times (-3) = 0 \), so 3 \( \ce{Mg^{2+}} \) and 2 \( \ce{N^{3-}} \).
- Lewis structure: \( [\ce{Mg^{2+}}]_3 \: \: [\ce{N^{3-}}]_2 \) (Mg has 0 valence electrons, each N has 8 dots, 3- charge).
General Format for Ion…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To draw the Lewis structures for these ionic compounds, we follow these general steps for ionic compounds (composed of cations and anions):
Step 1: Identify the ions
For an ionic compound \( M_xA_y \), the cation is \( M^{n+} \) and the anion is \( A^{m-} \), where \( n \) and \( m \) are the charges (determined by valence electrons or group number).
Step 2: Determine charges
- Group 1 metals (Li, Na, K, etc.): \( +1 \) charge.
- Group 2 metals (Mg, Ca, Ba, Sr, etc.): \( +2 \) charge.
- Group 13 metals (Al): \( +3 \) charge.
- Group 15 nonmetals (N, P, etc.): \( -3 \) charge (gain 3 electrons to fill octet).
- Group 16 nonmetals (O, S, Se, etc.): \( -2 \) charge (gain 2 electrons).
- Group 17 nonmetals (F, Cl, Br, etc.): \( -1 \) charge (gain 1 electron).
Example: \( \boldsymbol{\ce{Li2O}} \) (1)
- Li (Group 1): \( \ce{Li^+} \) (loses 1 electron).
- O (Group 16): \( \ce{O^{2-}} \) (gains 2 electrons).
- Charge balance: \( 2 \times (+1) + 1 \times (-2) = 0 \), so 2 \( \ce{Li^+} \) and 1 \( \ce{O^{2-}} \).
- Lewis structure: \( [\ce{Li^+}]_2 \: \: \ce{O^{2-}} \) (or draw each ion with valence electrons: Li has 0 valence electrons (lost 1), O has 8 (gained 2), so \( \ce{Li^+} \) (no dots) and \( \ce{O^{2-}} \) (8 dots, 2- charge)).
Example: \( \boldsymbol{\ce{MgS}} \) (2)
- Mg (Group 2): \( \ce{Mg^{2+}} \) (loses 2 electrons).
- S (Group 16): \( \ce{S^{2-}} \) (gains 2 electrons).
- Charge balance: \( 1 \times (+2) + 1 \times (-2) = 0 \), so 1 \( \ce{Mg^{2+}} \) and 1 \( \ce{S^{2-}} \).
- Lewis structure: \( \ce{Mg^{2+}} \: \: \ce{S^{2-}} \) (Mg has 0 valence electrons, S has 8 dots, 2- charge).
Example: \( \boldsymbol{\ce{K3N}} \) (3)
- K (Group 1): \( \ce{K^+} \) (loses 1 electron).
- N (Group 15): \( \ce{N^{3-}} \) (gains 3 electrons).
- Charge balance: \( 3 \times (+1) + 1 \times (-3) = 0 \), so 3 \( \ce{K^+} \) and 1 \( \ce{N^{3-}} \).
- Lewis structure: \( [\ce{K^+}]_3 \: \: \ce{N^{3-}} \) (K has 0 valence electrons, N has 8 dots, 3- charge).
Example: \( \boldsymbol{\ce{CaCl2}} \) (4)
- Ca (Group 2): \( \ce{Ca^{2+}} \) (loses 2 electrons).
- Cl (Group 17): \( \ce{Cl^-} \) (gains 1 electron).
- Charge balance: \( 1 \times (+2) + 2 \times (-1) = 0 \), so 1 \( \ce{Ca^{2+}} \) and 2 \( \ce{Cl^-} \).
- Lewis structure: \( \ce{Ca^{2+}} \: \: [\ce{Cl^-}]_2 \) (Ca has 0 valence electrons, each Cl has 8 dots, 1- charge).
Example: \( \boldsymbol{\ce{AlF3}} \) (6)
- Al (Group 13): \( \ce{Al^{3+}} \) (loses 3 electrons).
- F (Group 17): \( \ce{F^-} \) (gains 1 electron).
- Charge balance: \( 1 \times (+3) + 3 \times (-1) = 0 \), so 1 \( \ce{Al^{3+}} \) and 3 \( \ce{F^-} \).
- Lewis structure: \( \ce{Al^{3+}} \: \: [\ce{F^-}]_3 \) (Al has 0 valence electrons, each F has 8 dots, 1- charge).
Example: \( \boldsymbol{\ce{Al2S3}} \) (9)
- Al (Group 13): \( \ce{Al^{3+}} \) (loses 3 electrons).
- S (Group 16): \( \ce{S^{2-}} \) (gains 2 electrons).
- Charge balance: \( 2 \times (+3) + 3 \times (-2) = 0 \), so 2 \( \ce{Al^{3+}} \) and 3 \( \ce{S^{2-}} \).
- Lewis structure: \( [\ce{Al^{3+}}]_2 \: \: [\ce{S^{2-}}]_3 \) (Al has 0 valence electrons, each S has 8 dots, 2- charge).
Example: \( \boldsymbol{\ce{Mg3N2}} \) (10)
- Mg (Group 2): \( \ce{Mg^{2+}} \) (loses 2 electrons).
- N (Group 15): \( \ce{N^{3-}} \) (gains 3 electrons).
- Charge balance: \( 3 \times (+2) + 2 \times (-3) = 0 \), so 3 \( \ce{Mg^{2+}} \) and 2 \( \ce{N^{3-}} \).
- Lewis structure: \( [\ce{Mg^{2+}}]_3 \: \: [\ce{N^{3-}}]_2 \) (Mg has 0 valence electrons, each N has 8 dots, 3- charge).
General Format for Ionic Lewis Structures
For \( \ce{M^+_x A^-_{y}} \), draw the cation (no valence electrons, or \( \ce{M^+} \)) and the anion (with 8 valence electrons, \( \ce{A^-} \)) in the ratio given by the formula.
Final Answer (Example for \( \boldsymbol{\ce{Li2O}} \))
The Lewis structure for \( \ce{Li2O} \) is:
\( [\ce{Li^+}] \: [\ce{Li^+}] \: [\overset{..}{\underset{..}{\ce{O}}}]^{2-} \) (or simplified as \( [\ce{Li^+}]_2 \: \ce{O^{2-}} \) with the oxide ion showing 8 valence electrons).
For other compounds, follow the same logic (identify ions, balance charges, draw cations and anions with appropriate charges and valence electrons).