QUESTION IMAGE
Question
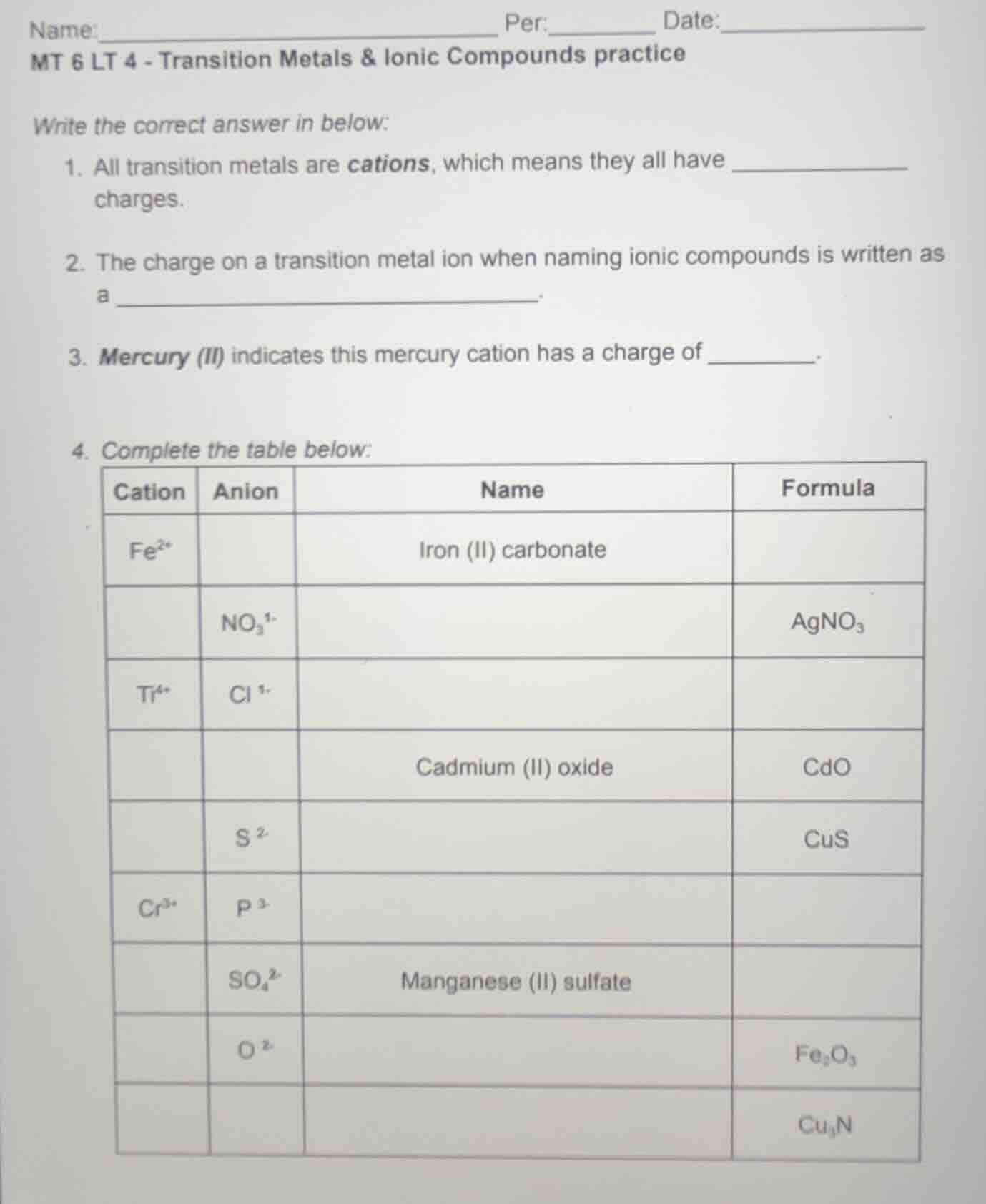
name:_____________________________ per:____ date:______________
mt 6 lt 4 - transition metals & ionic compounds practice
write the correct answer in below:
- all transition metals are cations, which means they all have _____________
charges.
- the charge on a transition metal ion when naming ionic compounds is written as
a _____________________________.
- mercury (ii) indicates this mercury cation has a charge of _________.
- complete the table below:
| cation | anion | name | formula |
|---|---|---|---|
| no₃¹⁻ | agno₃ | ||
| ti⁴⁺ | cl¹⁻ | ||
| cadmium (ii) oxide | cdo | ||
| s²⁻ | cus | ||
| cr³⁺ | p³⁻ | ||
| so₄²⁻ | manganese (ii) sulfate | ||
| o²⁻ | fe₂o₃ | ||
| cu₃n |
Question 1
Cations are ions with positive charges. Transition metals as cations have positive charges.
When naming ionic compounds with transition metals, the charge of the transition metal ion is written as a Roman numeral (in parentheses) to indicate its oxidation state.
The Roman numeral (II) after Mercury indicates the charge of the mercury cation. Roman numeral II corresponds to a charge of +2.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
positive