QUESTION IMAGE
Question
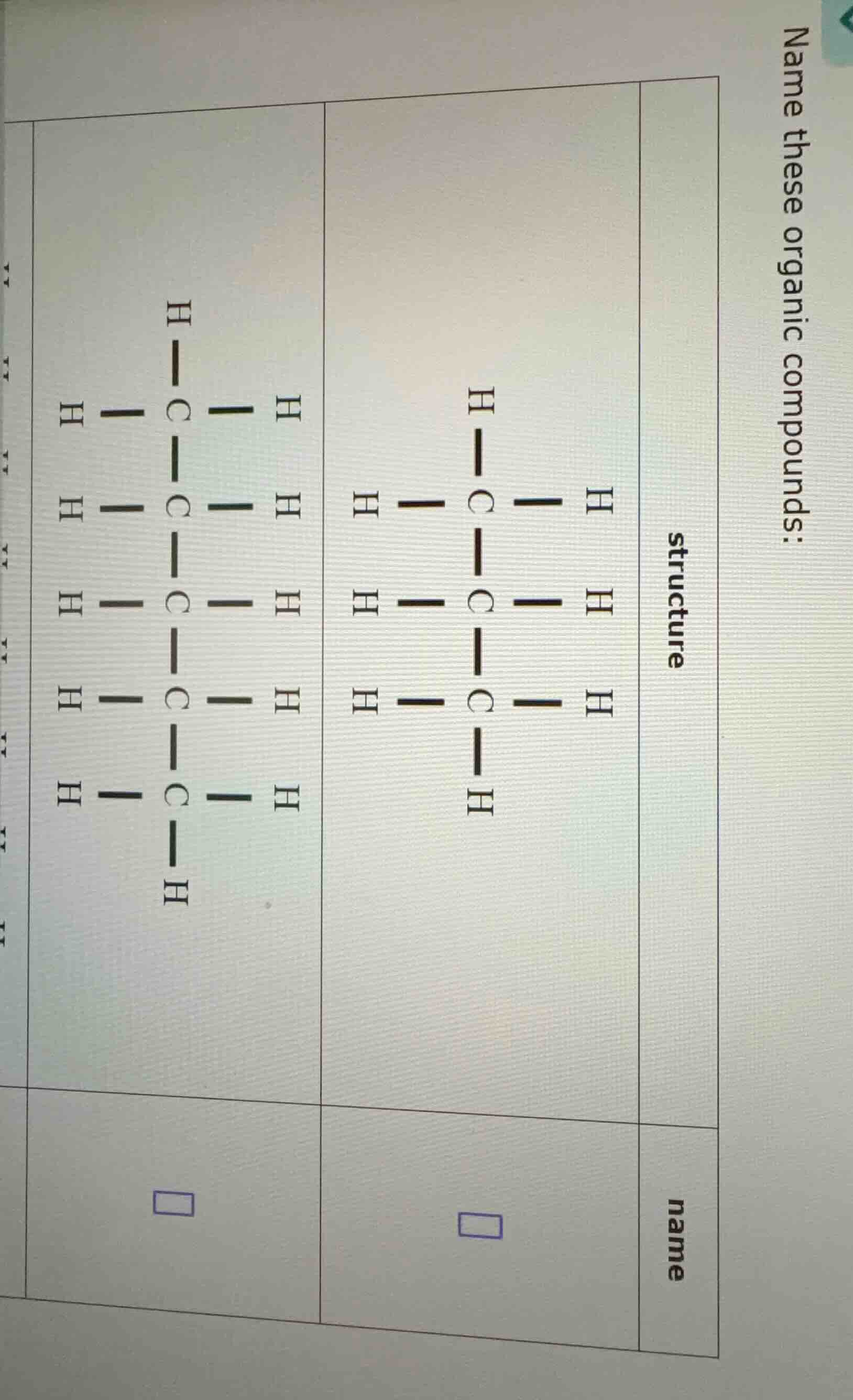
name these organic compounds:
structure
\
\
name
\square
\square
To name these organic compounds, we analyze their structures:
- First Structure (Right):
- The carbon chain has 3 carbon atoms (propane? Wait, no—wait, the structure is \( \text{H}_3\text{C}-\text{CH}_2-\text{CH}_3 \)? Wait, no, the given structure is a 3 - carbon chain? Wait, no, looking at the bonds: each carbon is bonded to hydrogens. Wait, the right structure: let's count carbons. The central carbon is bonded to two other carbons? Wait, no, the right structure: let's draw it. The structure is \( \text{H}-\text{C}(\text{H})-\text{C}(\text{H})-\text{H} \)? Wait, no, the right structure has 3 carbons? Wait, no, the right structure: the first (right) structure: let's count the carbon atoms. Wait, the right structure: the bonds are \( \text{H}-\text{C}-\text{C}-\text{H} \)? No, wait, the right structure is:
Wait, the right structure: each carbon is bonded as follows: the middle carbon is bonded to two carbons? No, wait, the right structure: let's count the number of carbon atoms. Wait, the right structure: the formula is \( \text{C}_3\text{H}_8 \)? No, wait, the right structure: let's see, the structure is \( \text{CH}_3-\text{CH}_2-\text{CH}_3 \)? No, wait, the right structure has 3 carbons? Wait, no, the right structure: the first (right) structure: the carbon chain is 3 carbons? Wait, no, the right structure: let's count the carbon atoms. Wait, the right structure: the bonds are \( \text{H}-\text{C}-\text{C}-\text{H} \)? No, the right structure is \( \text{CH}_3-\text{CH}_2-\text{CH}_3 \)? No, wait, the right structure is propane? Wait, no, the right structure: let's check the number of carbons. Wait, the right structure: the carbon chain is 3 carbons? Wait, no, the right structure: the formula is \( \text{C}_3\text{H}_8 \)? No, wait, the right structure: let's count the hydrogens. Each carbon: the first carbon (leftmost in the right structure) is bonded to 3 H, the middle to 2 H, the rightmost to 3 H? No, that can't be. Wait, the right structure: the correct structure is propane? Wait, no, the right structure is \( \text{CH}_3-\text{CH}_2-\text{CH}_3 \), which is propane. Wait, no, the right structure: let's look again. The right structure: the carbon chain is 3 carbons? Wait, no, the right structure: the bonds are \( \text{H}-\text{C}-\text{C}-\text{H} \) with the middle carbon bonded to two H? No, wait, the right structure is \( \text{CH}_3-\text{CH}_2-\text{CH}_3 \), which is propane (\( \text{C}_3\text{H}_8 \)). Wait, no, the right structure: let's count the carbon atoms. The right structure has 3 carbons? Wait, no, the right structure: the first (right) structure: the carbon chain is 3 carbons? Wait, no, the right structure: the formula is \( \text{C}_3\text{H}_8 \), which is propane. Wait, no, the right structure: let's check the left structure. The left structure has 6 carbons? Wait, the left structure: the carbon chain is 6 carbons? Wait, the left structure: let's count the carbon atoms. The left structure: the carbon chain is 6 carbons? Wait, the left structure: the formula is \( \text{C}_6\text{H}_{14} \), which is hexane. Wait, no, the left structure: let's count the carbon atoms. The left structure has 6 carbon atoms (since there are 6 C - C bonds in a straight chain). So the left structure is hexane (\( \text{C}_6\text{H}_{14} \)), and the right structure is propane (\( \text{C}_3\text{H}_8 \))? Wait, no, the right structure: let's count the carbon atoms. The right structure: the carbon chain is 3 carbons? Wait, no, the right structure: the bonds are \( \text{H}-\text{C}-\text{C}-\text{H} \) with the midd…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Right structure (3 - carbon chain): Propane
- Left structure (6 - carbon chain): Hexane
(Note: If the right structure is actually a 3 - carbon chain, it's propane (\( \text{C}_3\text{H}_8 \)); the left structure, with 6 carbon atoms in a straight chain, is hexane (\( \text{C}_6\text{H}_{14} \)).)