QUESTION IMAGE
Question
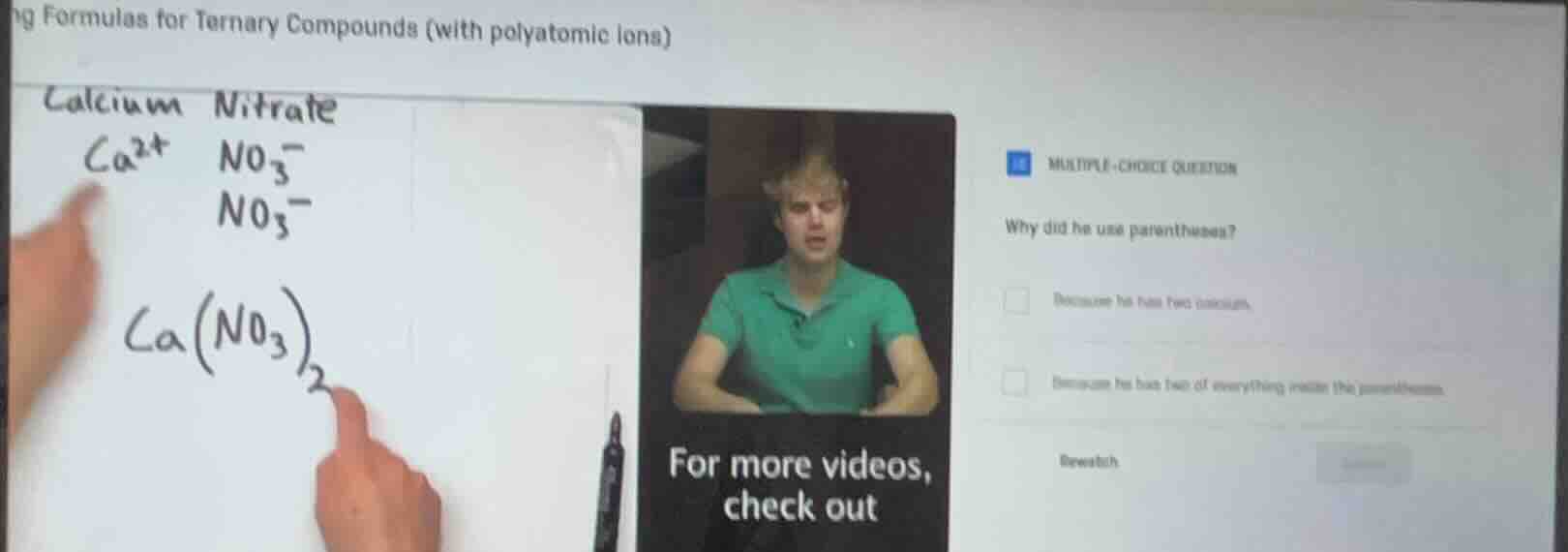
ng formulas for ternary compounds (with polyatomic lons)
calcium nitrate
ca²⁺ no₃⁻
no₃⁻
ca(no₃)₂
multiple-choice question
why did he use parentheses?
because he has two cations.
because he has two of everything inside the parentheses.
rewatch
To determine why parentheses are used in the formula \( \text{Ca(NO}_3\text{)}_2 \), we analyze the structure of the compound. Calcium has a charge of \( \text{Ca}^{2+} \), and the nitrate ion is \( \text{NO}_3^- \). To balance the charges, we need two nitrate ions for one calcium ion. Parentheses are used around the polyatomic ion \( \text{NO}_3^- \) when there is more than one of that polyatomic ion to indicate that the subscript (2, in this case) applies to the entire polyatomic ion. The first option ("Because he has two calciums") is incorrect because there is only one calcium ion. The second option ("Because he has two of everything inside the parentheses") is correct because the subscript 2 outside the parentheses means there are two nitrate ions (the entire polyatomic ion inside the parentheses is multiplied by 2).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Because he has two of everything inside the parentheses (assuming the second option is labeled B; adjust the label based on the actual question's option numbering, but the correct reasoning is that the subscript applies to the entire polyatomic ion inside the parentheses).