QUESTION IMAGE
Question
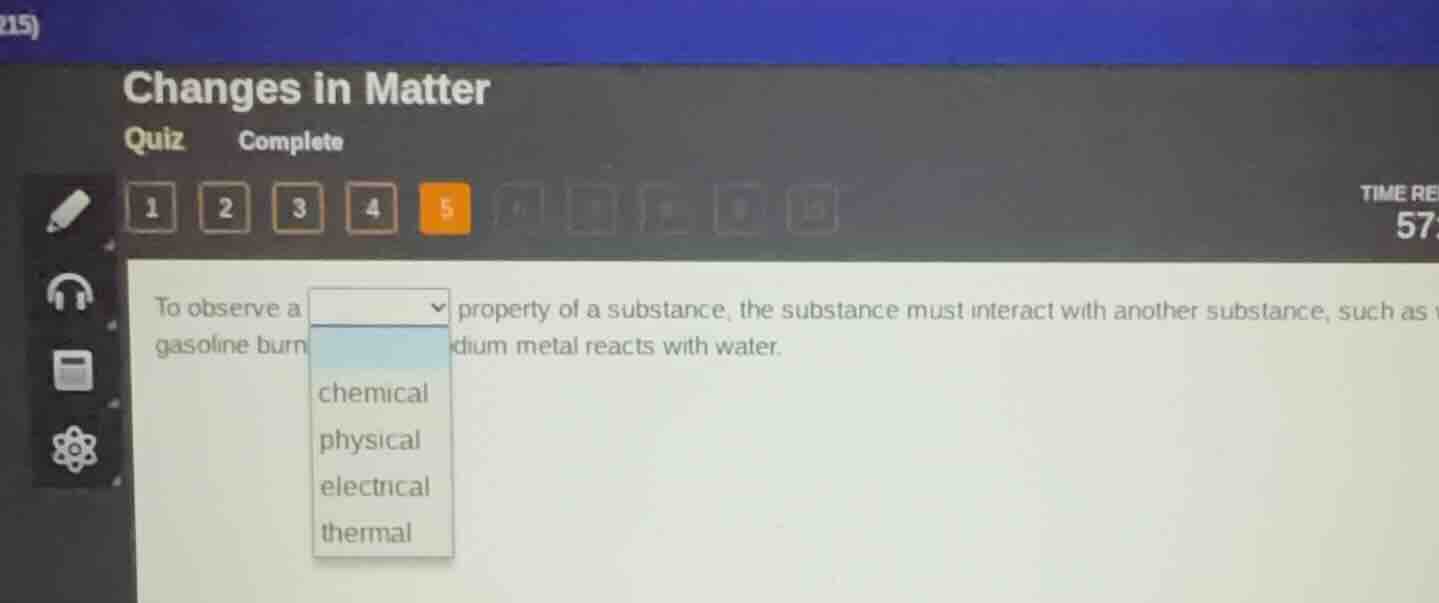
to observe a property of a substance, the substance must interact with another substance, such as gasoline burn dium metal reacts with water. options: chemical, physical, electrical, thermal
Chemical properties describe a substance's ability to undergo a chemical change or reaction with other substances. Examples like gasoline burning (reacting with oxygen) and sodium reacting with water are chemical changes that demonstrate chemical properties. Physical properties can be observed without altering the substance's chemical identity, while electrical/thermal properties relate to energy transfer, not chemical interaction with other substances.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
chemical