QUESTION IMAGE
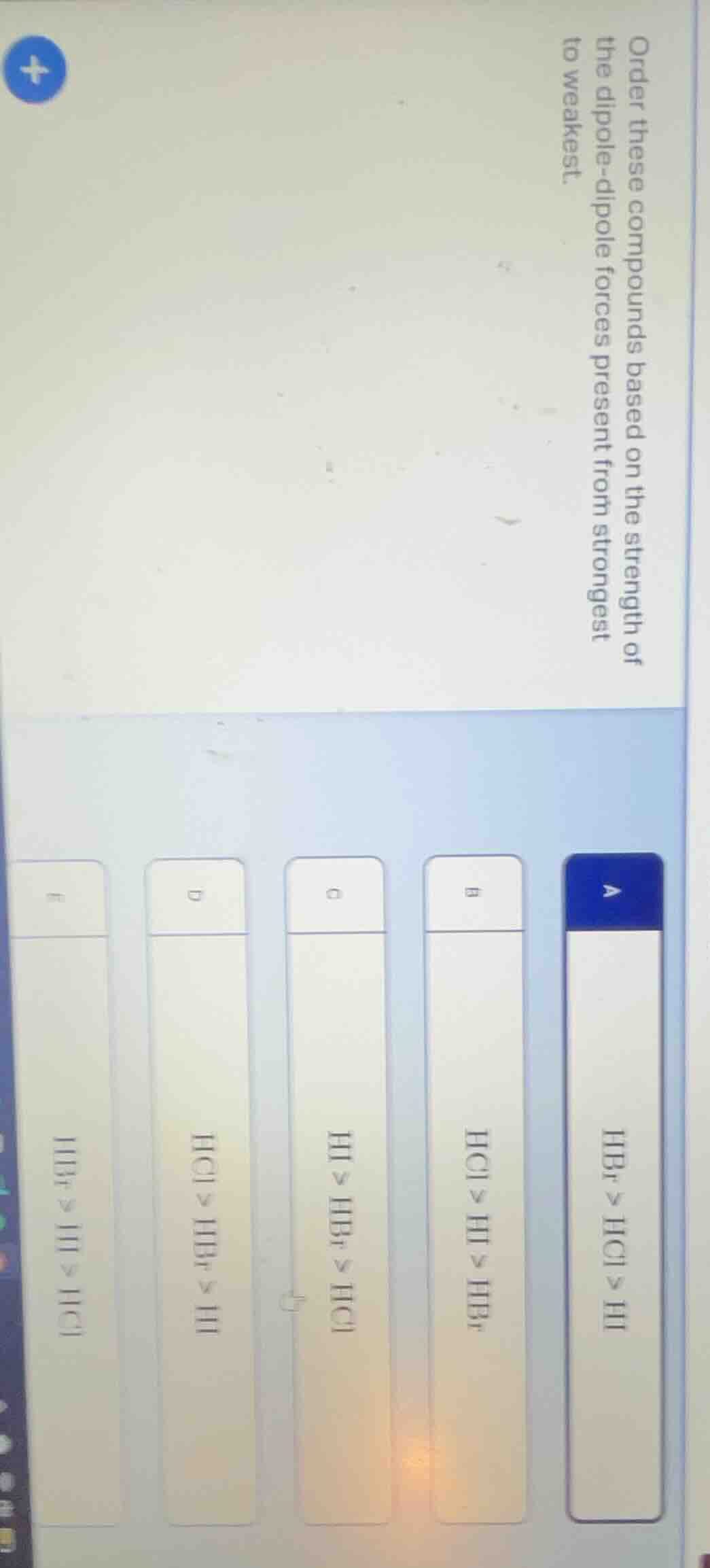
Question
order these compounds based on the strength of the dipole-dipole forces present from strongest to weakest.
a. hbr > hcl > hi
b. hcl > hi > hbr
c. hi > hbr > hcl
d. hcl > hbr > hi
e. hbr > hi > hcl
Step1: Relate polarity to dipole strength
Dipole-dipole force strength depends on molecular polarity, which is determined by the electronegativity difference ($\Delta EN$) between H and the halogen.
Step2: Calculate electronegativity differences
Electronegativities: $EN(\text{H})=2.2$, $EN(\text{Cl})=3.2$, $EN(\text{Br})=3.0$, $EN(\text{I})=2.7$
- $\Delta EN(\text{HCl}) = 3.2-2.2=1.0$
- $\Delta EN(\text{HBr}) = 3.0-2.2=0.8$
- $\Delta EN(\text{HI}) = 2.7-2.2=0.5$
Step3: Order by dipole force strength
Higher $\Delta EN$ = stronger dipole-dipole forces. So order: $\text{HCl} > \text{HBr} > \text{HI}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\text{HCl} > \text{HBr} > \text{HI}$