QUESTION IMAGE
Question
organic chemistry
loudon | parise seventh edition
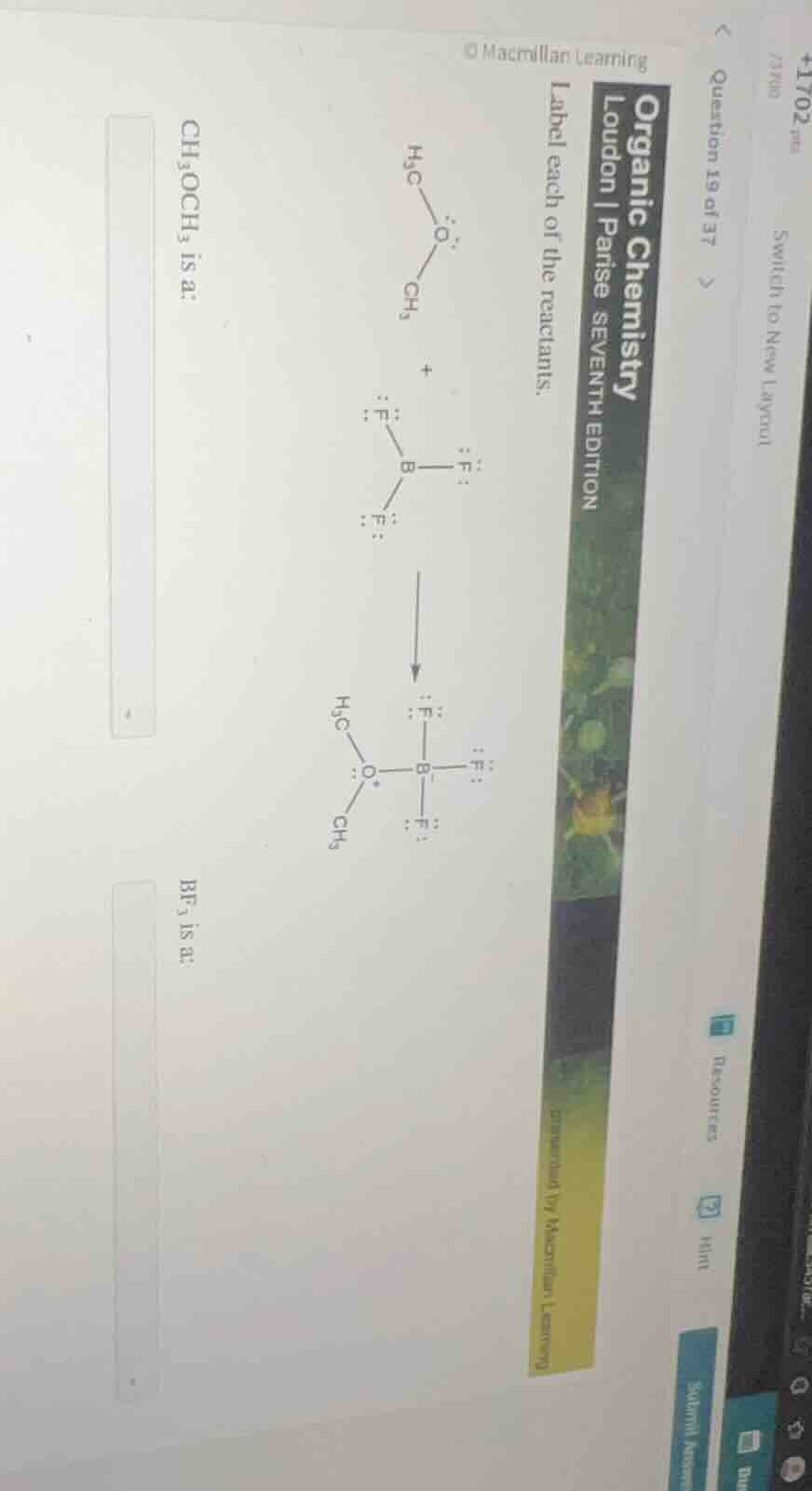
label each of the reactants.
$\ce{h3c - o - ch3}$ + $\ce{bf3}$ $\
ightarrow$ $\ce{h3c - o - b(f)3}$
$\ce{ch3och3}$ is a:
$\ce{bf3}$ is a:
In this Lewis acid-base reaction, $\text{CH}_3\text{OCH}_3$ (dimethyl ether) has a lone pair of electrons on the oxygen atom that it donates to form a bond with $\text{BF}_3$. A substance that donates an electron pair is a Lewis base. $\text{BF}_3$ has an incomplete octet on the boron atom, so it accepts the electron pair from the oxygen. A substance that accepts an electron pair is a Lewis acid.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{CH}_3\text{OCH}_3$ is a: Lewis base
$\text{BF}_3$ is a: Lewis acid