QUESTION IMAGE
Question
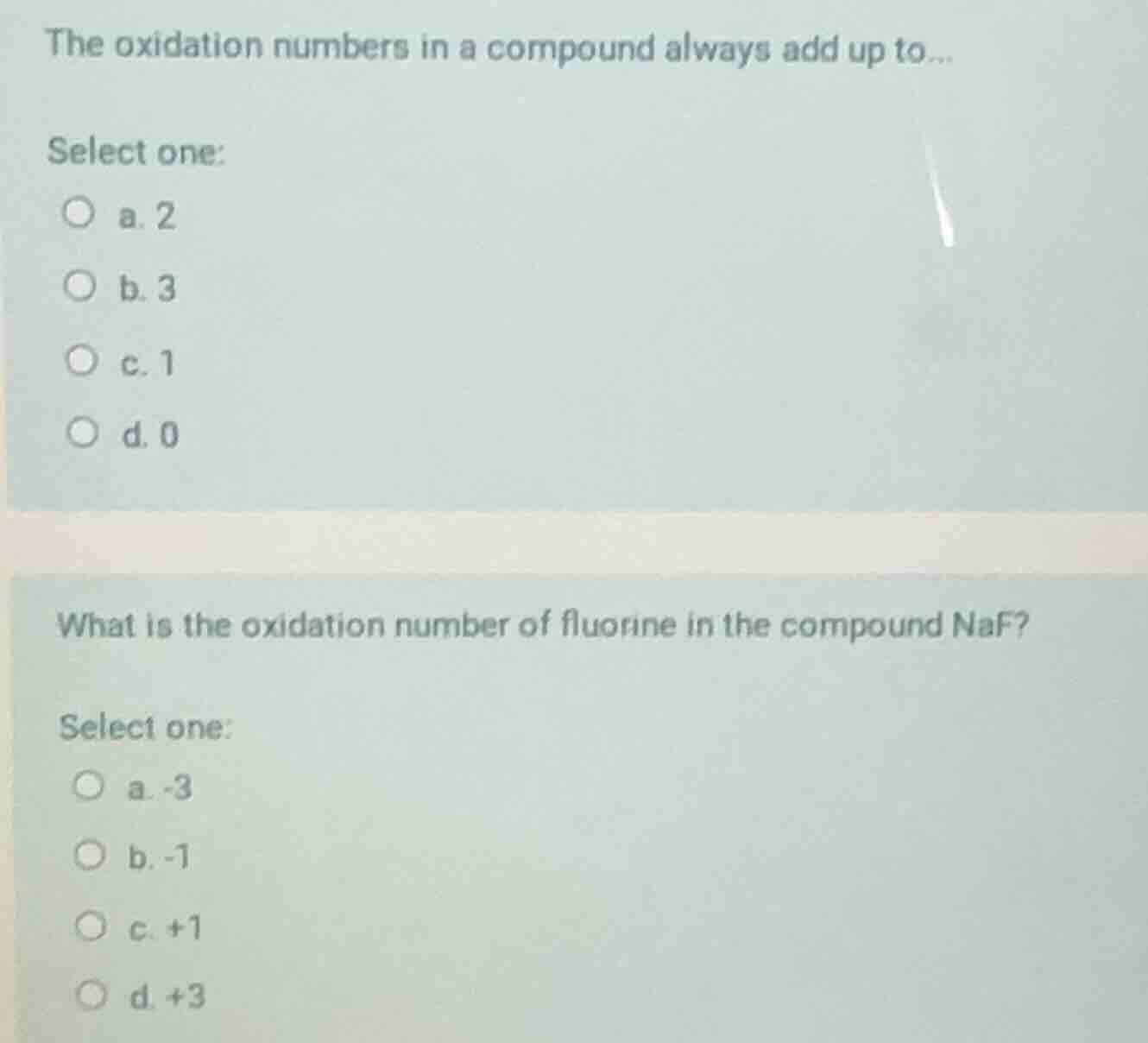
the oxidation numbers in a compound always add up to...
select one:
a. 2
b. 3
c. 1
d. 0
what is the oxidation number of fluorine in the compound naf?
select one:
a. -3
b. -1
c. +1
d. +3
Brief Explanations
For the first question, neutral compounds have no net electrical charge, so the sum of oxidation numbers of all atoms equals 0. For the second question, fluorine is the most electronegative element, so it always has an oxidation number of -1 in its compounds like NaF.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- d. 0
- b. -1