QUESTION IMAGE
Question
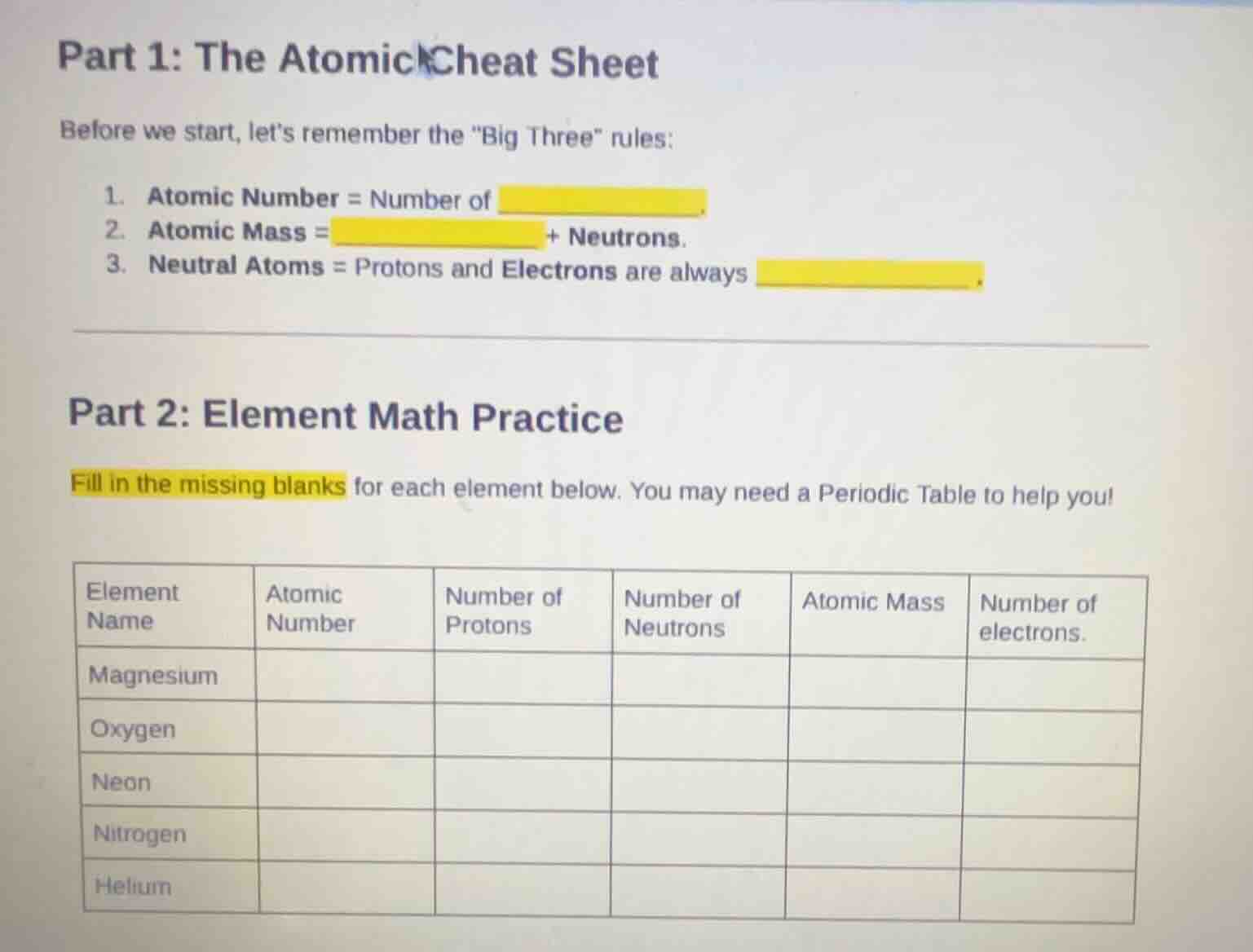
part 1: the atomic cheat sheet
before we start, lets remember the \big three\ rules:
- atomic number = number of
- atomic mass = + neutrons.
- neutral atoms = protons and electrons are always
part 2: element math practice
fill in the missing blanks for each element below. you may need a periodic table to help you!
| element name | atomic number | number of protons | number of neutrons | atomic mass | number of electrons. | --- | --- | --- | --- | --- | --- | magnesium | oxygen | neon | nitrogen | helium |
Step1: Fill "Big Three" rules
- Atomic Number = Number of $\text{Protons}$
- Atomic Mass = $\text{Protons}$ + Neutrons
- Neutral Atoms = Protons and Electrons are always $\text{equal}$
Step2: Get Magnesium data
From periodic table: Atomic Number = 12. Protons = Atomic Number = 12. Electrons = Protons = 12. Atomic Mass = 24.305, round to 24. Neutrons = $24 - 12 = 12$.
Step3: Get Oxygen data
From periodic table: Atomic Number = 8. Protons = 8. Electrons = 8. Atomic Mass = 16.00, round to 16. Neutrons = $16 - 8 = 8$.
Step4: Get Neon data
From periodic table: Atomic Number = 10. Protons = 10. Electrons = 10. Atomic Mass = 20.18, round to 20. Neutrons = $20 - 10 = 10$.
Step5: Get Nitrogen data
From periodic table: Atomic Number = 7. Protons = 7. Electrons = 7. Atomic Mass = 14.01, round to 14. Neutrons = $14 - 7 = 7$.
Step6: Get Helium data
From periodic table: Atomic Number = 2. Protons = 2. Electrons = 2. Atomic Mass = 4.00, round to 4. Neutrons = $4 - 2 = 2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 1 Completed Rules:
- Atomic Number = Number of $\text{Protons}$
- Atomic Mass = $\text{Protons}$ + Neutrons
- Neutral Atoms = Protons and Electrons are always $\text{equal}$
Part 2 Completed Table:
| Element Name | Atomic Number | Number of Protons | Number of Neutrons | Atomic Mass | Number of electrons |
|---|---|---|---|---|---|
| Oxygen | 8 | 8 | 8 | 16 | 8 |
| Neon | 10 | 10 | 10 | 20 | 10 |
| Nitrogen | 7 | 7 | 7 | 14 | 7 |
| Helium | 2 | 2 | 2 | 4 | 2 |