QUESTION IMAGE
Question
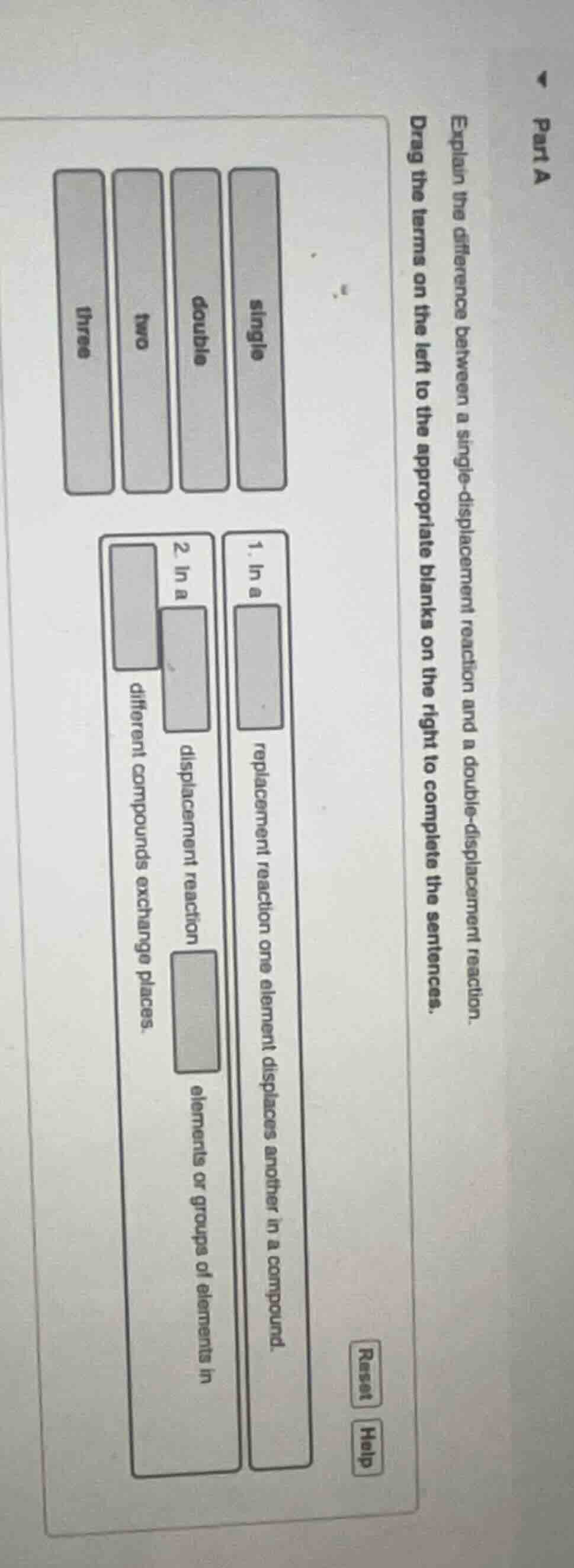
part a
explain the difference between a single - displacement reaction and a double - displacement reaction.
drag the terms on the left to the appropriate blanks on the right to complete the sentences.
terms: single, double, two, three
- in a \boxed{} replacement reaction one element displaces another in a compound.
- in a \boxed{} displacement reaction \boxed{} elements or groups of elements in \boxed{} different compounds exchange places.
(there are also reset and help buttons shown in the interface.)
Brief Explanations
- For the first sentence, a single - displacement reaction is defined as one element displacing another in a compound. So we use the term "single".
- For the second sentence, a double - displacement reaction involves two different compounds exchanging places (or two elements or groups of elements in two different compounds exchanging places). So we use the terms "double" and "two".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- In a $\boldsymbol{\text{single}}$ replacement reaction one element displaces another in a compound.
- In a $\boldsymbol{\text{double}}$ displacement reaction $\boldsymbol{\text{two}}$ elements or groups of elements in different compounds exchange places.