QUESTION IMAGE
Question
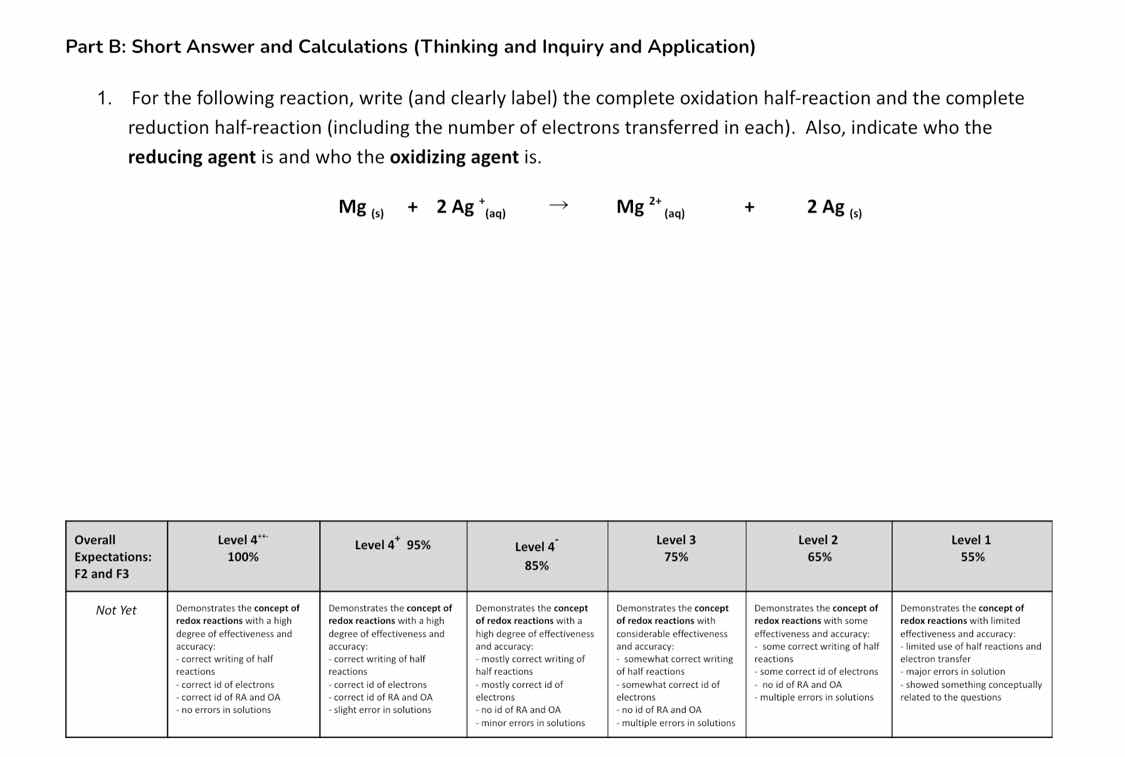
part b: short answer and calculations (thinking and inquiry and application)
- for the following reaction, write (and clearly label) the complete oxidation half - reaction and the complete reduction half - reaction (including the number of electrons transferred in each). also, indicate who the reducing agent is and who the oxidizing agent is.
\ce{mg_{(s)} + 2ag^{+}_{(aq)} -> mg^{2+}_{(aq)} + 2ag_{(s)}}
| overall expectations: f2 and f3 | not yet | level 4++ 100% | level 4+ 95% | level 4 85% | level 3 75% | level 2 65% | level 1 55% |
|---|
Step1: Identify Oxidation Half - Reaction
Oxidation is the loss of electrons. In the reaction \(Mg_{(s)}+2Ag^{+}_{(aq)}
ightarrow Mg^{2 + }_{(aq)}+2Ag_{(s)}\), magnesium (\(Mg\)) goes from an oxidation state of 0 (in \(Mg_{(s)}\)) to + 2 (in \(Mg^{2+}_{(aq)}\)). So the oxidation half - reaction is the process where \(Mg\) loses electrons. The oxidation half - reaction is: \(Mg_{(s)}
ightarrow Mg^{2+}_{(aq)} + 2e^-\). Here, \(Mg\) loses 2 electrons.
Step2: Identify Reduction Half - Reaction
Reduction is the gain of electrons. Silver (\(Ag\)) goes from an oxidation state of + 1 (in \(Ag^{+}_{(aq)}\)) to 0 (in \(Ag_{(s)}\)). For 2 moles of \(Ag^{+}\), the reduction half - reaction is: \(2Ag^{+}_{(aq)}+2e^-
ightarrow 2Ag_{(s)}\). Here, each \(Ag^{+}\) gains 1 electron, and for 2 \(Ag^{+}\) ions, a total of 2 electrons are gained.
Step3: Identify Oxidizing and Reducing Agents
The reducing agent is the substance that is oxidized (loses electrons). Since \(Mg\) is oxidized (loses electrons in the oxidation half - reaction), \(Mg\) is the reducing agent. The oxidizing agent is the substance that is reduced (gains electrons). Since \(Ag^{+}\) is reduced (gains electrons in the reduction half - reaction), \(Ag^{+}\) (or the species containing \(Ag^{+}\), in this case, the \(Ag^{+}_{(aq)}\) from the reactants) is the oxidizing agent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Oxidation half - reaction: \(Mg_{(s)}
ightarrow Mg^{2+}_{(aq)} + 2e^-\) (oxidation, 2 electrons lost)
- Reduction half - reaction: \(2Ag^{+}_{(aq)}+2e^-
ightarrow 2Ag_{(s)}\) (reduction, 2 electrons gained)
- Reducing agent: \(Mg_{(s)}\) (because it is oxidized, loses electrons)
- Oxidizing agent: \(Ag^{+}_{(aq)}\) (because it is reduced, gains electrons)