QUESTION IMAGE
Question
pebworth, logan
sps_physical science_checkpoint2_2025-2026
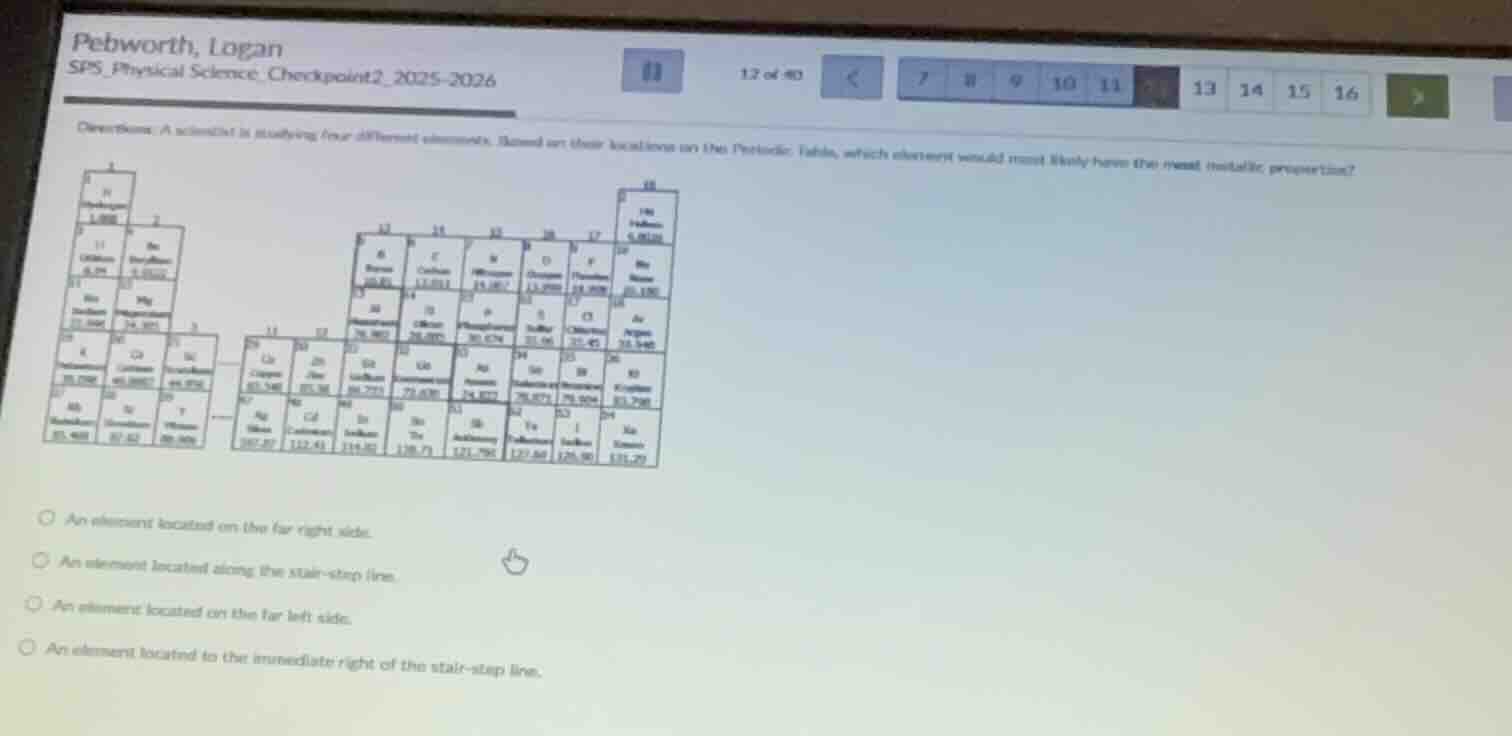
question: a scientist is studying four different elements. based on their locations on the periodic table, which element would most likely have the most metallic properties?
periodic table image
options:
- an element located on the far right side.
- an element located along the stair - step line.
- an element located on the far left side.
- an element located to the immediate right of the stair - step line.
Metallic properties increase as you move left and down on the periodic table. Elements on the far left are alkali and alkaline earth metals, which have the highest metallic character. Elements on the far right are nonmetals, those along the stair-step line are metalloids, and elements right of the stair-step are nonmetals with low metallic properties.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
An element located on the far left side.