QUESTION IMAGE
Question
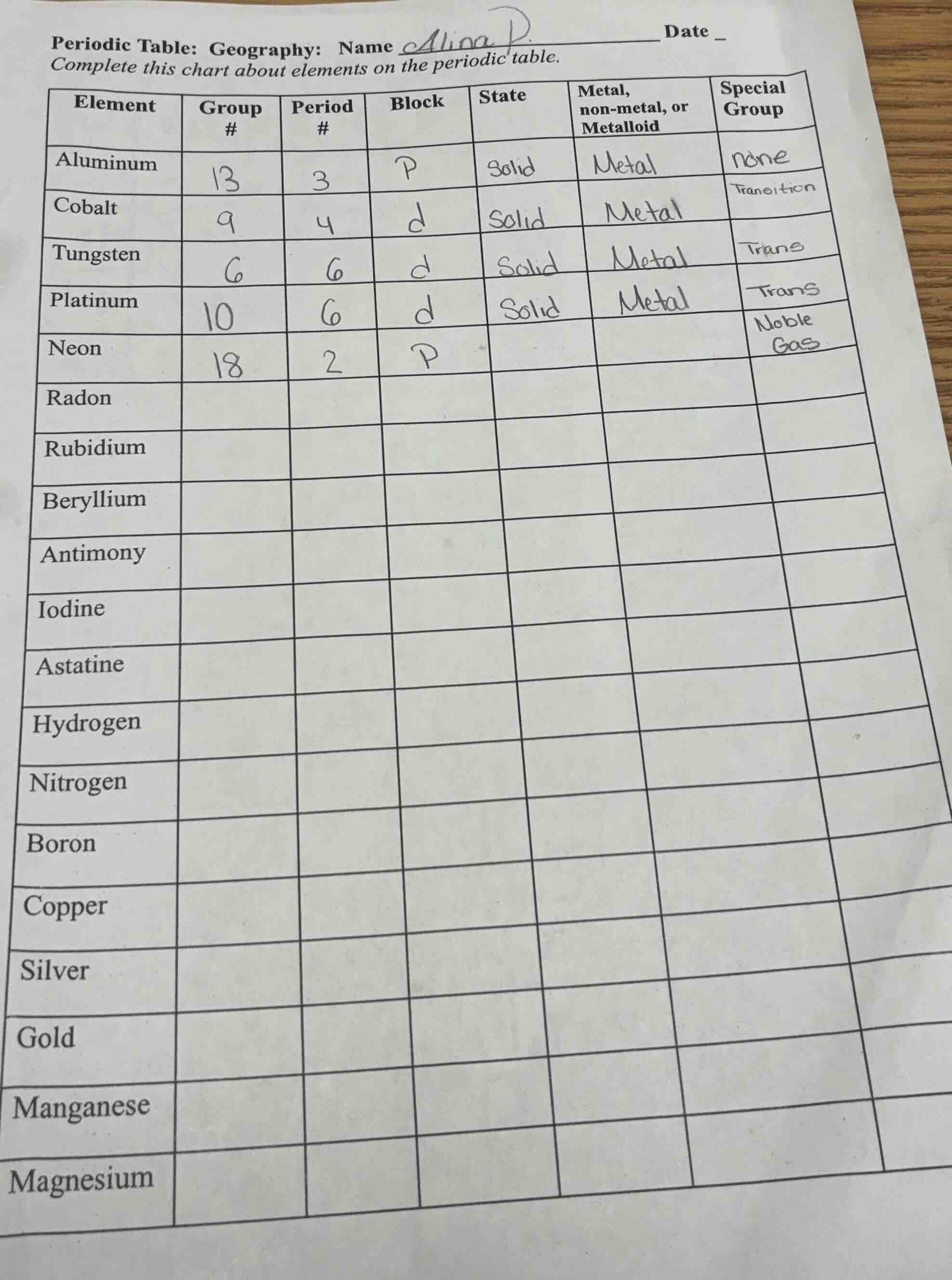
periodic table: geography: name alina p. date
complete this chart about elements on the periodic table.
element | group # | period # | block | state | metal, non - metal, or metalloid | special group
aluminum | 13 | 3 | p | solid | metal | none
cobalt | 9 | 4 | d | solid | metal | transition
tungsten | 6 | 6 | d | solid | metal | trans
platinum | 10 | 6 | d | solid | metal | trans
neon | 18 | 2 | p | | | noble gas
radon | | | | | |
rubidium | | | | | |
beryllium | | | | | |
antimony | | | | | |
iodine | | | | | |
astatine | | | | | |
hydrogen | | | | | |
nitrogen | | | | | |
boron | | | | | |
copper | | | | | |
silver | | | | | |
gold | | | | | |
manganese | | | | | |
magnesium | | | | | |
To complete the chart, we use the periodic table's properties (group, period, block, state, metal/non - metal/metalloid, special group) for each element:
Radon
- Group #: 18 (noble gases are in group 18)
- Period #: 6 (radon is in period 6)
- Block: p (noble gases are in p - block)
- State: Gas (radon is a gaseous element at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (noble gases are non - metals)
- Special Group: Noble Gas (it belongs to the noble gas group)
Rubidium
- Group #: 1 (alkali metals are in group 1)
- Period #: 5 (rubidium is in period 5)
- Block: s (alkali metals are in s - block)
- State: Solid (rubidium is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (alkali metals are metals)
- Special Group: Alkali Metal (it belongs to the alkali metal group)
Beryllium
- Group #: 2 (alkaline earth metals are in group 2)
- Period #: 2 (beryllium is in period 2)
- Block: s (alkaline earth metals are in s - block)
- State: Solid (beryllium is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (alkaline earth metals are metals)
- Special Group: Alkaline Earth Metal (it belongs to the alkaline earth metal group)
Antimony
- Group #: 15 (pnictogens are in group 15)
- Period #: 5 (antimony is in period 5)
- Block: p (antimony is in p - block)
- State: Solid (antimony is a solid metalloid at room temperature)
- Metal, non - metal, or Metalloid: Metalloid (antimony has properties of both metals and non - metals)
- Special Group: None (no special named group other than pnictogen, but often "none" is used if not a common special group like alkali, noble gas etc.)
Iodine
- Group #: 17 (halogens are in group 17)
- Period #: 5 (iodine is in period 5)
- Block: p (halogens are in p - block)
- State: Solid (iodine is a solid non - metal at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (halogens are non - metals)
- Special Group: Halogen (it belongs to the halogen group)
Astatine
- Group #: 17 (halogens are in group 17)
- Period #: 6 (astatine is in period 6)
- Block: p (halogens are in p - block)
- State: Solid (astatine is a solid non - metal/metalloid at room temperature, mostly considered non - metal)
- Metal, non - metal, or Metalloid: Non - metal (or Metalloid, but more commonly non - metal)
- Special Group: Halogen (it belongs to the halogen group)
Hydrogen
- Group #: 1 (it is in group 1)
- Period #: 1 (hydrogen is in period 1)
- Block: s (hydrogen is in s - block)
- State: Gas (hydrogen is a gaseous non - metal at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (hydrogen is a non - metal)
- Special Group: None (unique, not part of alkali metals as it's a non - metal)
Nitrogen
- Group #: 15 (pnictogens are in group 15)
- Period #: 2 (nitrogen is in period 2)
- Block: p (nitrogen is in p - block)
- State: Gas (nitrogen is a gaseous non - metal at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (nitrogen is a non - metal)
- Special Group: None (no special named group other than pnictogen, but often "none" is used)
Boron
- Group #: 13 (boron group is in group 13)
- Period #: 2 (boron is in period 2)
- Block: p (boron is in p - block)
- State: Solid (boron is a solid metalloid at room temperature)
- Metal, non - metal, or Metalloid: Metalloid (boron has propertie…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To complete the chart, we use the periodic table's properties (group, period, block, state, metal/non - metal/metalloid, special group) for each element:
Radon
- Group #: 18 (noble gases are in group 18)
- Period #: 6 (radon is in period 6)
- Block: p (noble gases are in p - block)
- State: Gas (radon is a gaseous element at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (noble gases are non - metals)
- Special Group: Noble Gas (it belongs to the noble gas group)
Rubidium
- Group #: 1 (alkali metals are in group 1)
- Period #: 5 (rubidium is in period 5)
- Block: s (alkali metals are in s - block)
- State: Solid (rubidium is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (alkali metals are metals)
- Special Group: Alkali Metal (it belongs to the alkali metal group)
Beryllium
- Group #: 2 (alkaline earth metals are in group 2)
- Period #: 2 (beryllium is in period 2)
- Block: s (alkaline earth metals are in s - block)
- State: Solid (beryllium is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (alkaline earth metals are metals)
- Special Group: Alkaline Earth Metal (it belongs to the alkaline earth metal group)
Antimony
- Group #: 15 (pnictogens are in group 15)
- Period #: 5 (antimony is in period 5)
- Block: p (antimony is in p - block)
- State: Solid (antimony is a solid metalloid at room temperature)
- Metal, non - metal, or Metalloid: Metalloid (antimony has properties of both metals and non - metals)
- Special Group: None (no special named group other than pnictogen, but often "none" is used if not a common special group like alkali, noble gas etc.)
Iodine
- Group #: 17 (halogens are in group 17)
- Period #: 5 (iodine is in period 5)
- Block: p (halogens are in p - block)
- State: Solid (iodine is a solid non - metal at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (halogens are non - metals)
- Special Group: Halogen (it belongs to the halogen group)
Astatine
- Group #: 17 (halogens are in group 17)
- Period #: 6 (astatine is in period 6)
- Block: p (halogens are in p - block)
- State: Solid (astatine is a solid non - metal/metalloid at room temperature, mostly considered non - metal)
- Metal, non - metal, or Metalloid: Non - metal (or Metalloid, but more commonly non - metal)
- Special Group: Halogen (it belongs to the halogen group)
Hydrogen
- Group #: 1 (it is in group 1)
- Period #: 1 (hydrogen is in period 1)
- Block: s (hydrogen is in s - block)
- State: Gas (hydrogen is a gaseous non - metal at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (hydrogen is a non - metal)
- Special Group: None (unique, not part of alkali metals as it's a non - metal)
Nitrogen
- Group #: 15 (pnictogens are in group 15)
- Period #: 2 (nitrogen is in period 2)
- Block: p (nitrogen is in p - block)
- State: Gas (nitrogen is a gaseous non - metal at room temperature)
- Metal, non - metal, or Metalloid: Non - metal (nitrogen is a non - metal)
- Special Group: None (no special named group other than pnictogen, but often "none" is used)
Boron
- Group #: 13 (boron group is in group 13)
- Period #: 2 (boron is in period 2)
- Block: p (boron is in p - block)
- State: Solid (boron is a solid metalloid at room temperature)
- Metal, non - metal, or Metalloid: Metalloid (boron has properties of both metals and non - metals)
- Special Group: None (no special named group other than boron group, but often "none" is used)
Copper
- Group #: 11 (coinage metals are in group 11)
- Period #: 4 (copper is in period 4)
- Block: d (copper is in d - block)
- State: Solid (copper is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (copper is a metal)
- Special Group: Transition Metal (it is a transition metal)
Silver
- Group #: 11 (coinage metals are in group 11)
- Period #: 5 (silver is in period 5)
- Block: d (silver is in d - block)
- State: Solid (silver is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (silver is a metal)
- Special Group: Transition Metal (it is a transition metal)
Gold
- Group #: 11 (coinage metals are in group 11)
- Period #: 6 (gold is in period 6)
- Block: d (gold is in d - block)
- State: Solid (gold is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (gold is a metal)
- Special Group: Transition Metal (it is a transition metal)
Manganese
- Group #: 7 (manganese is in group 7)
- Period #: 4 (manganese is in period 4)
- Block: d (manganese is in d - block)
- State: Solid (manganese is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (manganese is a metal)
- Special Group: Transition Metal (it is a transition metal)
Magnesium
- Group #: 2 (alkaline earth metals are in group 2)
- Period #: 3 (magnesium is in period 3)
- Block: s (alkaline earth metals are in s - block)
- State: Solid (magnesium is a solid metal at room temperature)
- Metal, non - metal, or Metalloid: Metal (magnesium is a metal)
- Special Group: Alkaline Earth Metal (it belongs to the alkaline earth metal group)
Now, let's tabulate the answers:
| Element | Group # | Period # | Block | State | Metal, non - metal, or Metalloid | Special Group |
|---|---|---|---|---|---|---|
| Rubidium | 1 | 5 | s | Solid | Metal | Alkali Metal |
| Beryllium | 2 | 2 | s | Solid | Metal | Alkaline Earth Metal |
| Antimony | 15 | 5 | p | Solid | Metalloid | None |
| Iodine | 17 | 5 | p | Solid | Non - metal | Halogen |
| Astatine | 17 | 6 | p | Solid | Non - metal | Halogen |
| Hydrogen | 1 | 1 | s | Gas | Non - metal | None |
| Nitrogen | 15 | 2 | p | Gas | Non - metal | None |
| Boron | 13 | 2 | p | Solid | Metalloid | None |
| Copper | 11 | 4 | d | Solid | Metal | Transition Metal |
| Silver | 11 | 5 | d | Solid | Metal | Transition Metal |
| Gold | 11 | 6 | d | Solid | Metal | Transition Metal |
| Manganese | 7 | 4 | d | Solid | Metal | Transition Metal |
| Magnesium | 2 | 3 | s | Solid | Metal | Alkaline Earth Metal |