QUESTION IMAGE
Question
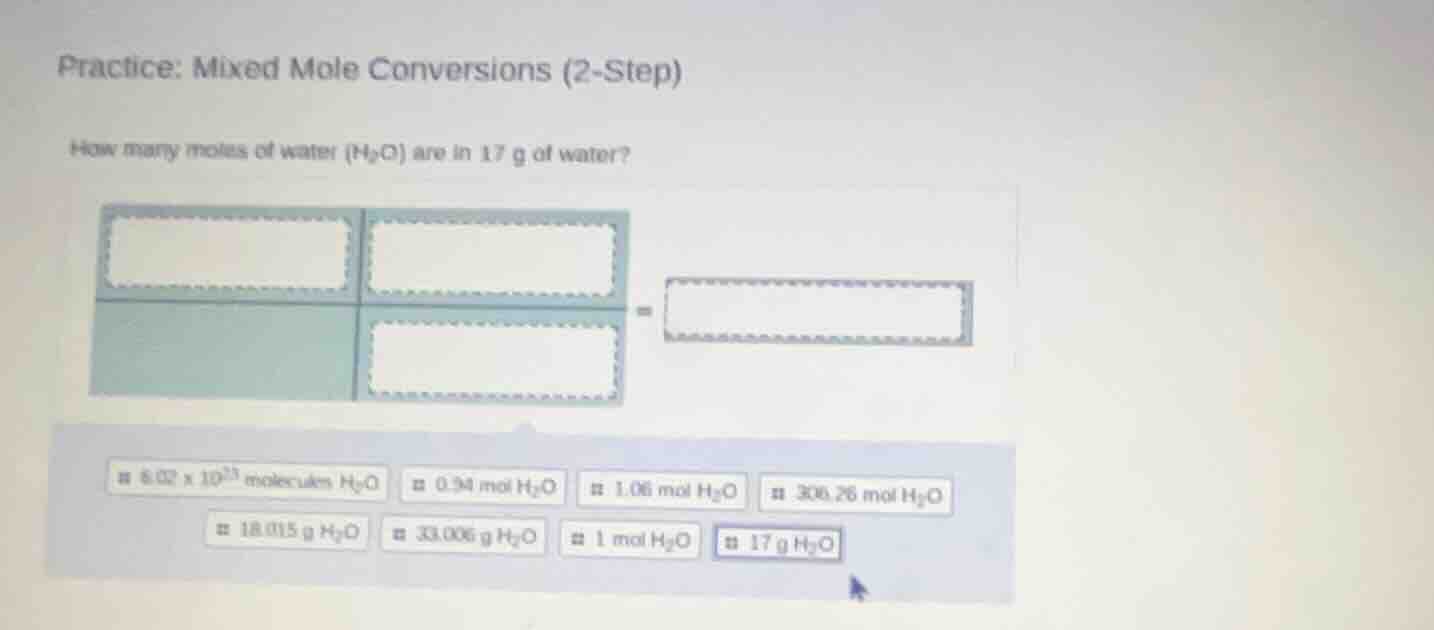
practice: mixed mole conversions (2-step)
how many moles of water (h₂o) are in 17 g of water?
8.02 × 10²³ molecules h₂o 0.94 mol h₂o 1.06 mol h₂o 306.26 mol h₂o
18.015 g h₂o 33.006 g h₂o 1 mol h₂o 17 g h₂o
Step1: Recall molar mass of \(H_2O\)
Molar mass of \(H_2O\) is calculated as \(2\times1.008 + 16.00 = 18.016\space g/mol\) (approx \(18.015\space g/mol\) as given).
Step2: Use mass - mole conversion formula
The formula to convert mass to moles is \(n=\frac{m}{M}\), where \(n\) is moles, \(m\) is mass, and \(M\) is molar mass.
Given \(m = 17\space g\) and \(M=18.015\space g/mol\), so \(n=\frac{17\space g}{18.015\space g/mol}\approx0.9437\space mol\)? Wait, no, wait the options have \(0.94\)? Wait the options: let's check the calculation again. Wait \(17\div18.015\approx0.943\), but the options have \(0.94\space mol\) (the second option: \(0.94\space mol\space H_2O\)). Wait, maybe I miscalculated? Wait \(17\div18.015\approx0.943\), which is approximately \(0.94\space mol\).
Wait, let's do the calculation step by step with the given values. The mass of water is \(17\space g\), molar mass of water is \(18.015\space g/mol\). The number of moles \(n=\frac{mass}{molar\space mass}=\frac{17\space g}{18.015\space g/mol}\approx0.94\space mol\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(\boldsymbol{0.94\space mol\space H_2O}\) (corresponding to the option: \(\boldsymbol{\square\ 0.94\space mol\ H_2O}\))