QUESTION IMAGE
Question
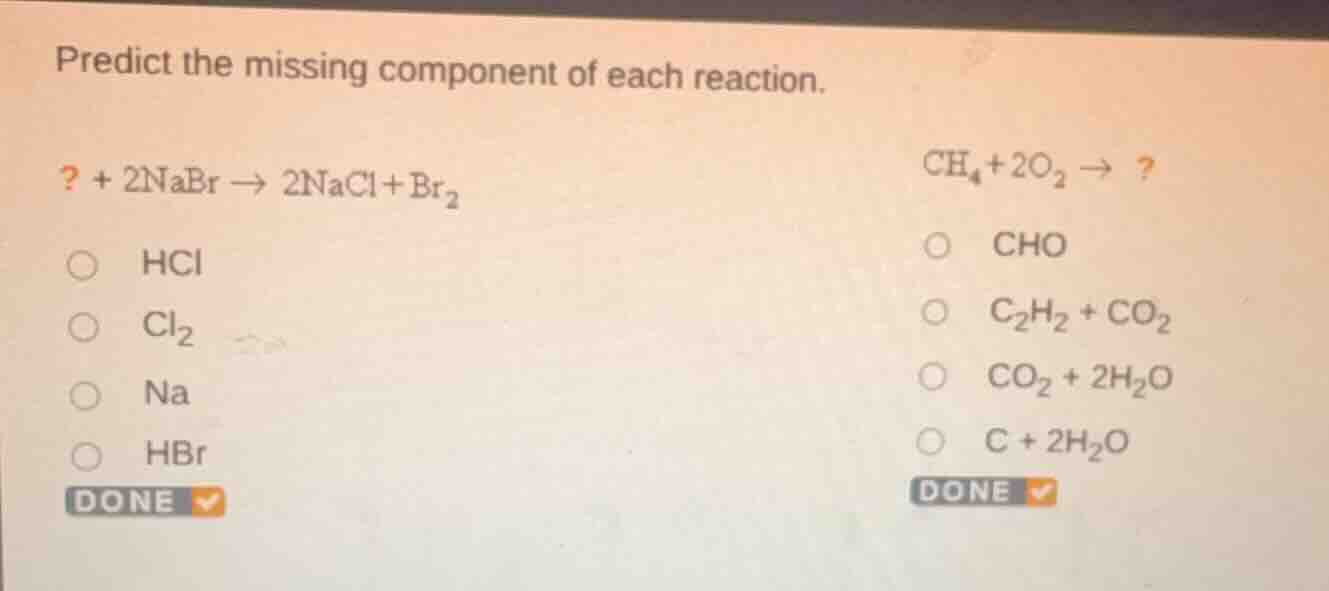
predict the missing component of each reaction.
? + 2nabr → 2nacl + br₂
hcl
cl₂
na
hbr
ch₄ + 2o₂ → ?
cho
c₂h₂ + co₂
co₂ + 2h₂o
c + 2h₂o
First Reaction ( ? + 2NaBr → 2NaCl + Br₂ )
Step1: Analyze Reactants and Products
We know that in a chemical reaction, the number of atoms of each element should be conserved on both sides. Let's check the elements: Na, Br, Cl. On the product side, we have 2NaCl (so 2 Na, 2 Cl) and Br₂ (2 Br). On the reactant side, we have 2NaBr (2 Na, 2 Br). So we need a reactant that provides 2 Cl.
Step2: Evaluate Options
- HCl: If we use HCl, the reaction would involve H, but there's no H in products. So no.
- Cl₂: Cl₂ has 2 Cl atoms. Let's check the reaction: Cl₂ + 2NaBr → 2NaCl + Br₂. This balances (2 Cl, 2 Na, 2 Br on both sides).
- Na: Na would add more Na, but we already have enough Na from NaBr. No.
- HBr: HBr has H and Br, but products have no H. No.
So the missing reactant is Cl₂.
Second Reaction ( CH₄ + 2O₂ → ? )
Step1: Recognize Reaction Type
CH₄ (methane) reacting with O₂ is a combustion reaction. In combustion of hydrocarbons, the products are usually CO₂ and H₂O.
Step2: Evaluate Options
- CHO: Not a valid product of methane combustion.
- C₂H₂ + CO₂: Not a product of CH₄ combustion.
- CO₂ + 2H₂O: Let's check atoms. CH₄ has 1 C, 4 H. 2O₂ has 4 O. Products: CO₂ (1 C, 2 O) and 2H₂O (4 H, 2 O). Total O: 2 + 2 = 4 (matches 2O₂). C: 1 (matches CH₄). H: 4 (matches CH₄).
- C + 2H₂O: Not a product of complete combustion of methane.
So the product is CO₂ + 2H₂O.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First reaction: $\boldsymbol{\text{Cl}_2}$ (from the options: $\boldsymbol{\text{Cl}_2}$)
Second reaction: $\boldsymbol{\text{CO}_2 + 2\text{H}_2\text{O}}$ (from the options: $\boldsymbol{\text{CO}_2 + 2\text{H}_2\text{O}}$)