QUESTION IMAGE
Question
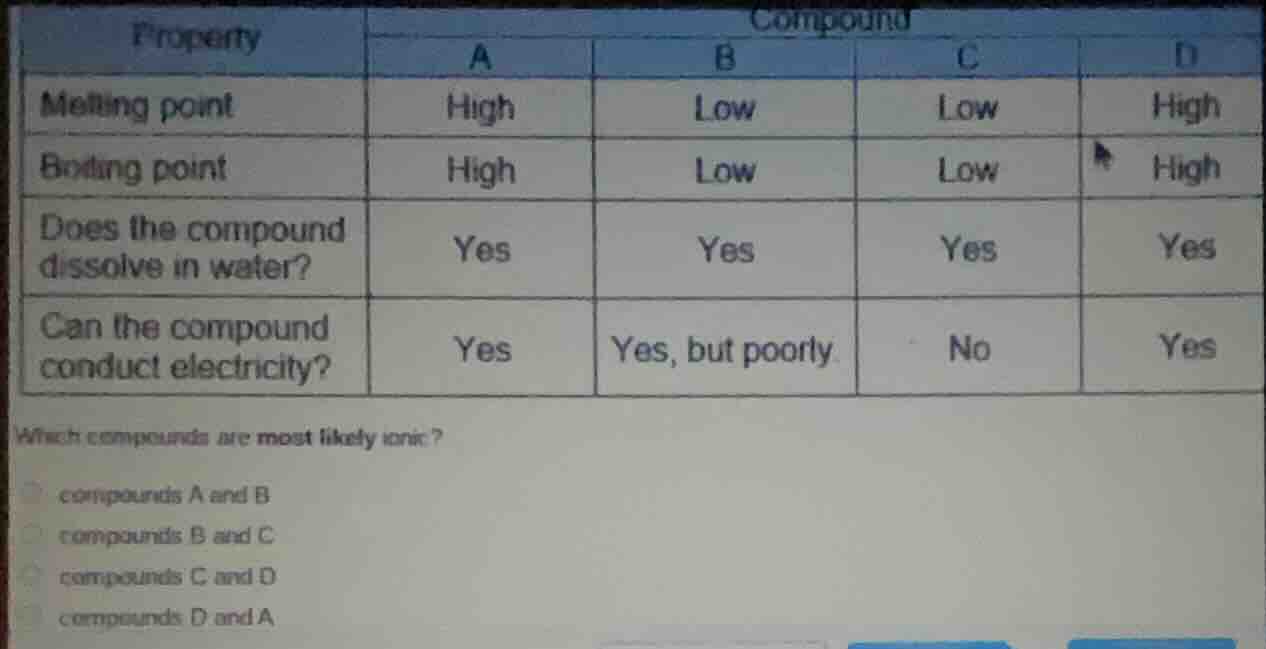
property compound a b c d melting point high low low high boiling point high low low high does the compound dissolve in water? yes yes yes yes can the compound conduct electricity? yes yes, but poorly no yes which compounds are most likely ionic? compounds a and b compounds b and c compounds c and d compounds d and a
Step1: Recall ionic compound properties
Ionic compounds typically have high melting/boiling points, dissolve in water, and conduct electricity in aqueous form.
Step2: Match compounds to properties
Compound A: High m.p./b.p., dissolves in water, conducts electricity → matches ionic properties.
Compound B: Low m.p./b.p., conducts poorly → does not match ionic properties.
Compound C: Low m.p./b.p., does not conduct electricity → does not match ionic properties.
Compound D: High m.p./b.p., dissolves in water, conducts electricity → matches ionic properties.
Step3: Identify correct option
Compounds A and D fit the ionic compound profile, which corresponds to the last option.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
compounds D and A