QUESTION IMAGE
Question
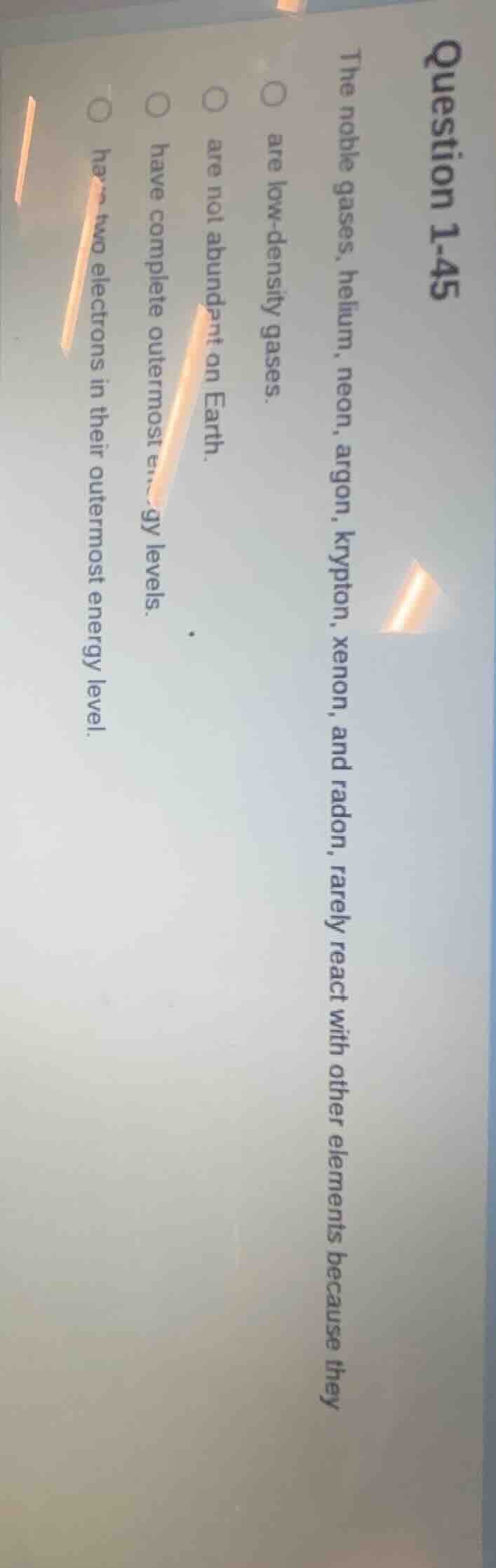
question 1-45
the noble gases, helium, neon, argon, krypton, xenon, and radon, rarely react with other elements because they
○ are low-density gases.
○ are not abundant on earth.
○ have complete outermost energy levels.
○ have two electrons in their outermost energy level.
Noble gases (helium, neon, argon, krypton, xenon, radon) rarely react because they have complete outermost energy levels (valence shells). Low - density, abundance on Earth, or two electrons (only helium has two, others have eight) don't explain their chemical inertness. Chemical reactivity is related to electron configuration, and noble gases have a stable, complete outer electron shell, so they don't easily form bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
have complete outermost energy levels.