QUESTION IMAGE
Question
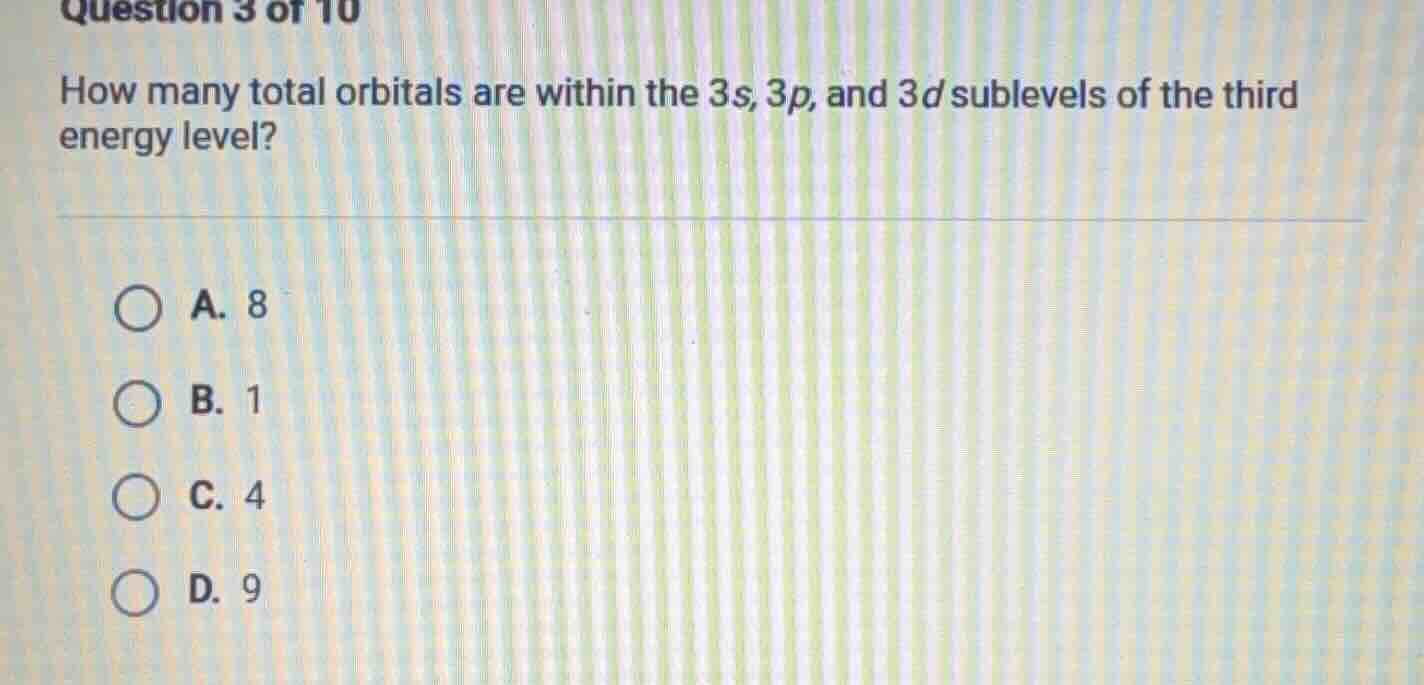
question 3 of 10
how many total orbitals are within the 3s, 3p, and 3d sublevels of the third energy level?
a. 8
b. 1
c. 4
d. 9
Step1: Recall orbital counts per sublevel
- \(s\) sublevel: 1 orbital (regardless of \(n\), here \(3s\) has 1 orbital).
- \(p\) sublevel: 3 orbitals (any \(np\) sublevel, so \(3p\) has 3 orbitals).
- \(d\) sublevel: 5 orbitals (any \(nd\) sublevel, so \(3d\) has 5 orbitals).
Step2: Sum the orbitals
Total orbitals = Orbitals in \(3s\) + Orbitals in \(3p\) + Orbitals in \(3d\)
Total orbitals = \(1 + 3 + 5\)
Total orbitals = \(9\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. 9