QUESTION IMAGE
Question
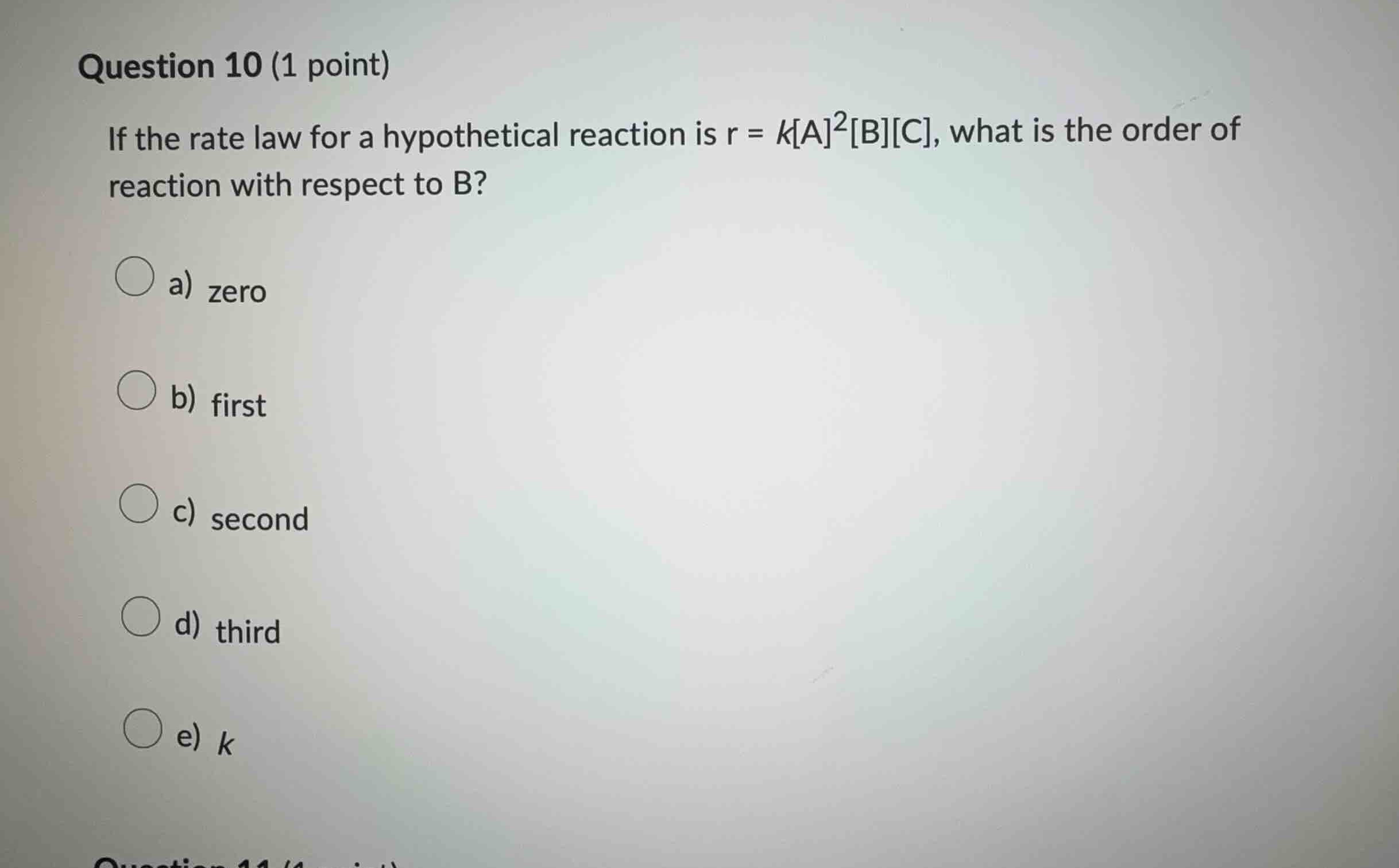
question 10 (1 point)
if the rate law for a hypothetical reaction is $r = k\text{a}^2\text{b}\text{c}$, what is the order of reaction with respect to b?
\bigcirc a) zero
\bigcirc b) first
\bigcirc c) second
\bigcirc d) third
\bigcirc e) $k$
Step1: Recall reaction order definition
The order of reaction with respect to a reactant is the exponent of its concentration term in the rate law.
Step2: Identify exponent of [B]
In the rate law \( r = k[\text{A}]^2[\text{B}][\text{C}] \), the concentration of B is raised to the power of 1 (since \( [\text{B}] = [\text{B}]^1 \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) first