QUESTION IMAGE
Question
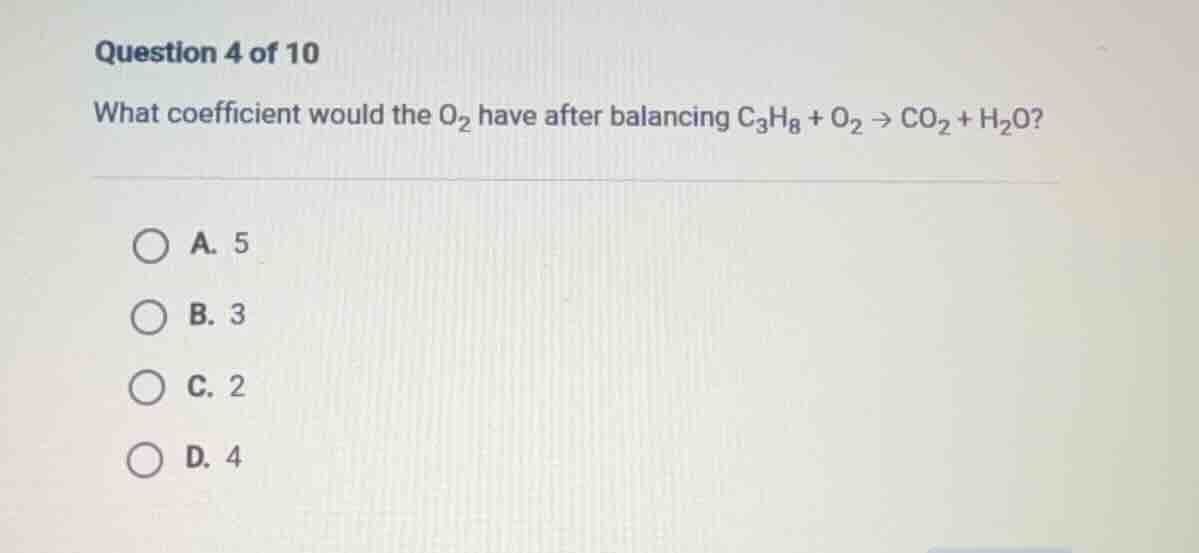
question 4 of 10
what coefficient would the o₂ have after balancing c₃h₈ + o₂ → co₂ + h₂o?
a. 5
b. 3
c. 2
d. 4
Step1: Balance Carbon atoms
In \( \text{C}_3\text{H}_8 \), there are 3 C atoms. So, we put a coefficient of 3 in front of \( \text{CO}_2 \) to balance C:
\( \text{C}_3\text{H}_8 + \text{O}_2
ightarrow 3\text{CO}_2 + \text{H}_2\text{O} \)
Step2: Balance Hydrogen atoms
In \( \text{C}_3\text{H}_8 \), there are 8 H atoms. Each \( \text{H}_2\text{O} \) has 2 H atoms, so we need \( \frac{8}{2} = 4 \) molecules of \( \text{H}_2\text{O} \):
\( \text{C}_3\text{H}_8 + \text{O}_2
ightarrow 3\text{CO}_2 + 4\text{H}_2\text{O} \)
Step3: Balance Oxygen atoms
On the right side, \( 3\text{CO}_2 \) has \( 3 \times 2 = 6 \) O atoms, and \( 4\text{H}_2\text{O} \) has \( 4 \times 1 = 4 \) O atoms. Total O on the right: \( 6 + 4 = 10 \). Each \( \text{O}_2 \) has 2 O atoms, so we need \( \frac{10}{2} = 5 \) molecules of \( \text{O}_2 \):
\( \text{C}_3\text{H}_8 + 5\text{O}_2
ightarrow 3\text{CO}_2 + 4\text{H}_2\text{O} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. 5