QUESTION IMAGE
Question
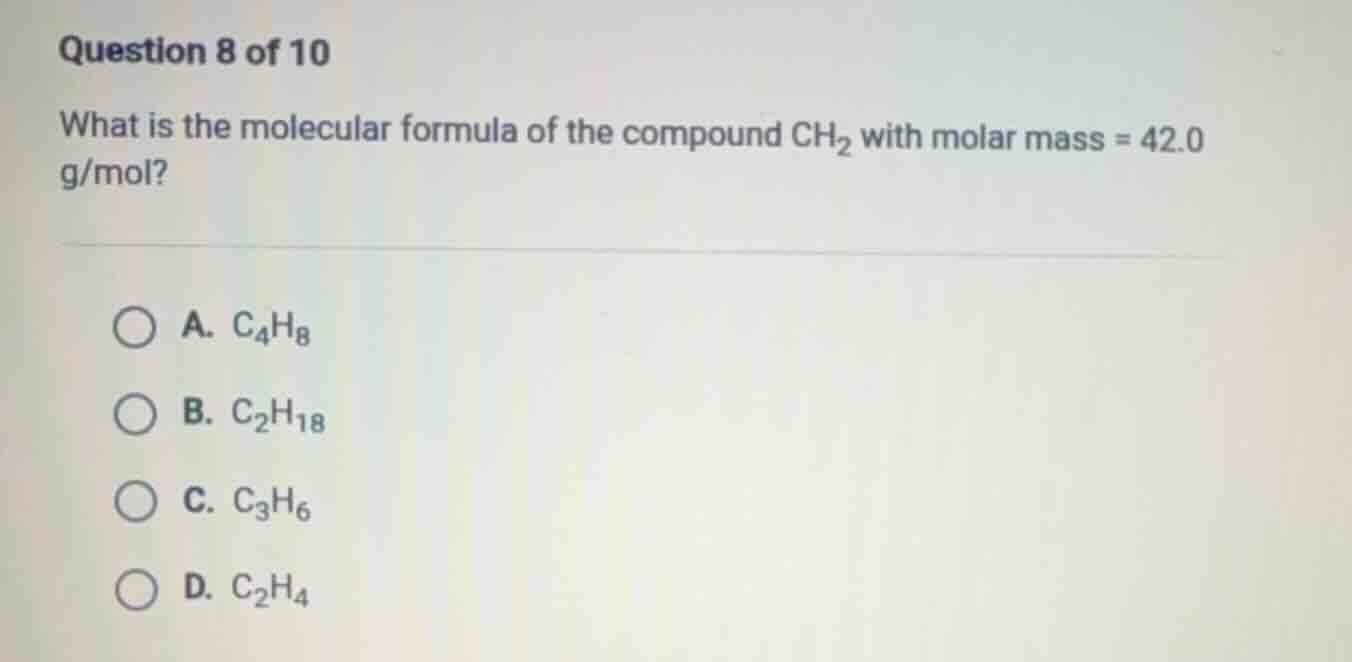
question 8 of 10
what is the molecular formula of the compound ch₂ with molar mass = 42.0 g/mol?
a. c₄h₈
b. c₂h₁₈
c. c₃h₆
d. c₂h₄
Step1: Calculate molar mass of empirical formula
The empirical formula is \( CH_2 \). Molar mass of \( C = 12.01 \, g/mol \), molar mass of \( H = 1.008 \, g/mol \). So molar mass of \( CH_2 \) is \( 12.01 + 2\times1.008 = 14.026 \, g/mol \).
Step2: Find the multiplier \( n \)
\( n=\frac{\text{Molar mass of molecular formula}}{\text{Molar mass of empirical formula}}=\frac{42.0}{14.026}\approx3 \).
Step3: Determine molecular formula
Multiply subscripts in empirical formula (\( CH_2 \)) by \( n = 3 \). So molecular formula is \( C_{3}H_{6} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \( C_3H_6 \)