QUESTION IMAGE
Question
question 4 of 14
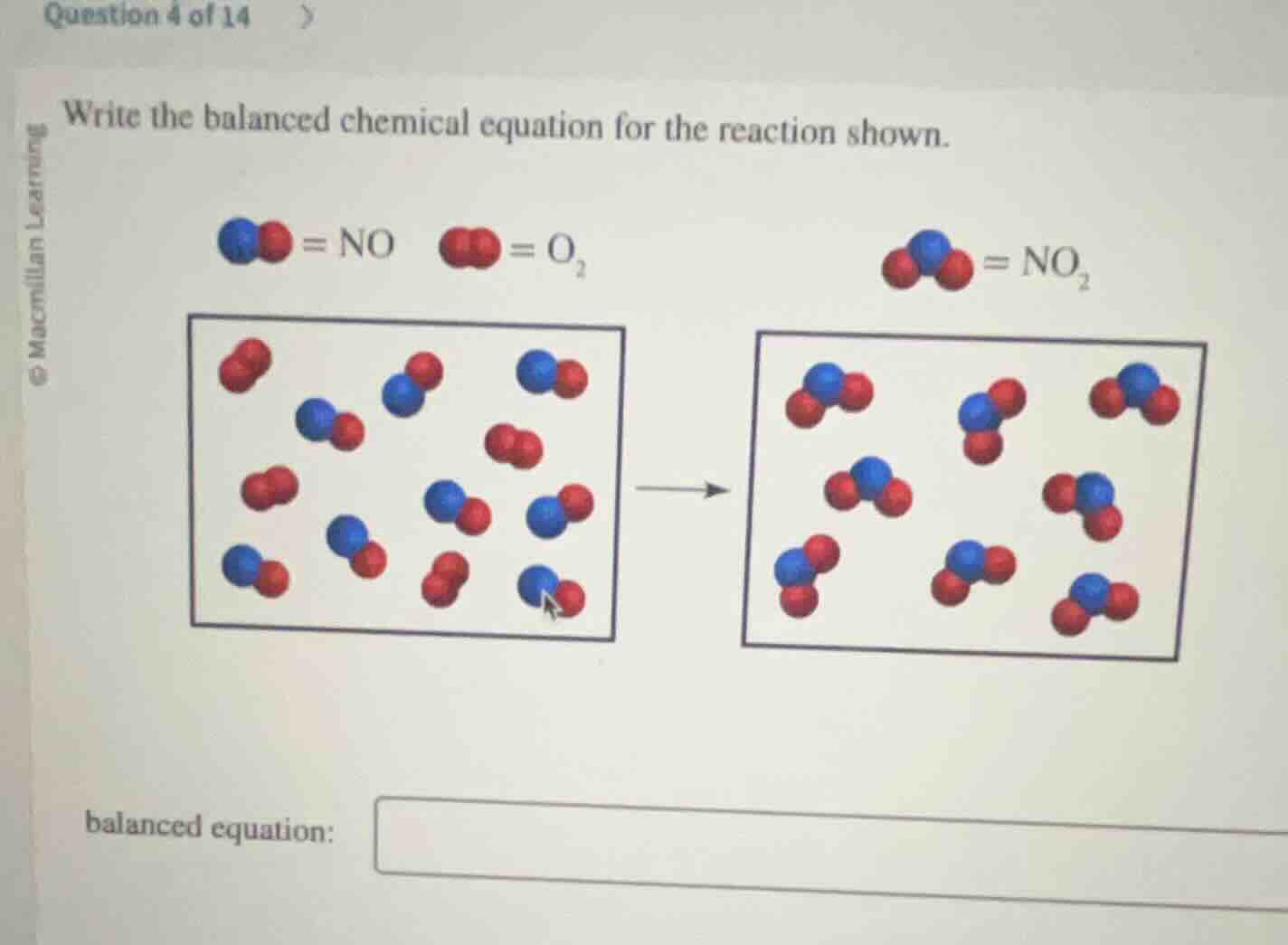
write the balanced chemical equation for the reaction shown.
= no = o₂ = no₂
balanced equation:
Step1: Count reactant molecules
Reactants: 8 NO, 4 O₂
Step2: Count product molecules
Products: 8 NO₂
Step3: Write unbalanced equation
$\text{NO} + \text{O}_2
ightarrow \text{NO}_2$
Step4: Balance atoms (N then O)
First balance N: 2 NO + O₂ → 2 NO₂. Verify: 2 N on left/right; 2+2=4 O on left, 4 O on right. Scale to match the diagram counts (multiply by 4): $8\text{NO} + 4\text{O}_2 = 8\text{NO}_2$, then simplify to the smallest whole number ratio by dividing all coefficients by 4.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{2\text{NO} + \text{O}_2 = 2\text{NO}_2}$