QUESTION IMAGE
Question
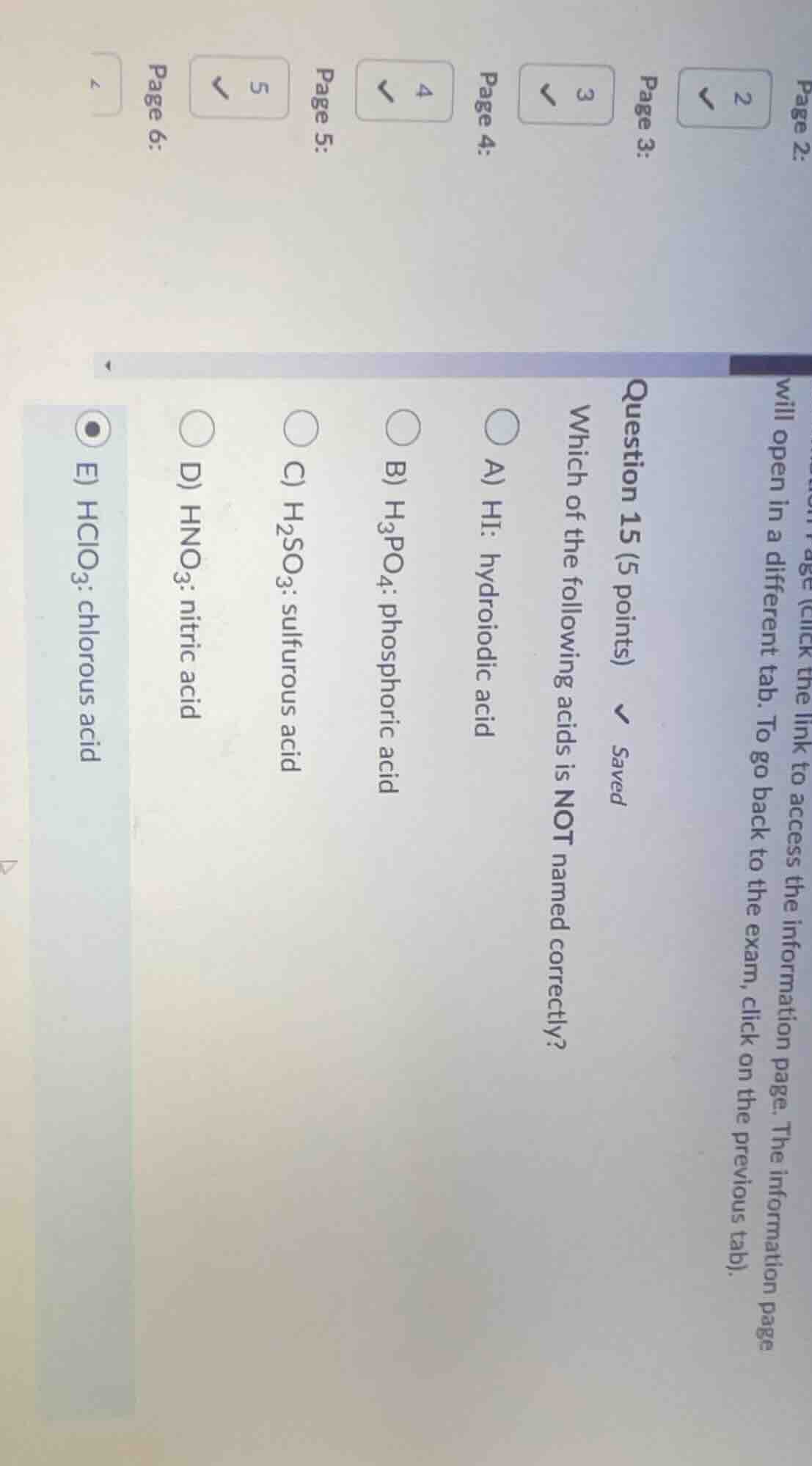
question 15 (5 points) saved which of the following acids is not named correctly? a) hi: hydroiodic acid b) h₃po₄: phosphoric acid c) h₂so₃: sulfurous acid d) hno₃: nitric acid e) hclo₃: chlorous acid
Brief Explanations
To determine the incorrectly named acid, we analyze each option:
- Option A: HI is hydroiodic acid (correct, as binary acids use "hydro - root - ic acid").
- Option B: \(H_3PO_4\) is phosphoric acid (correct, \(PO_4^{3 - }\) is phosphate, so the acid is phosphoric).
- Option C: \(H_2SO_3\) is sulfurous acid (correct, \(SO_3^{2 - }\) is sulfite, so the acid is sulfurous).
- Option D: \(HNO_3\) is nitric acid (correct, \(NO_3^-\) is nitrate, so the acid is nitric).
- Option E: \(HClO_3\) should be chloric acid, not chlorous acid. Chlorous acid is \(HClO_2\) (since \(ClO_2^-\) is chlorite, \(ClO_3^-\) is chlorate, so the acid from \(ClO_3^-\) is chloric acid).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
E) HClO₃: chlorous acid