QUESTION IMAGE
Question
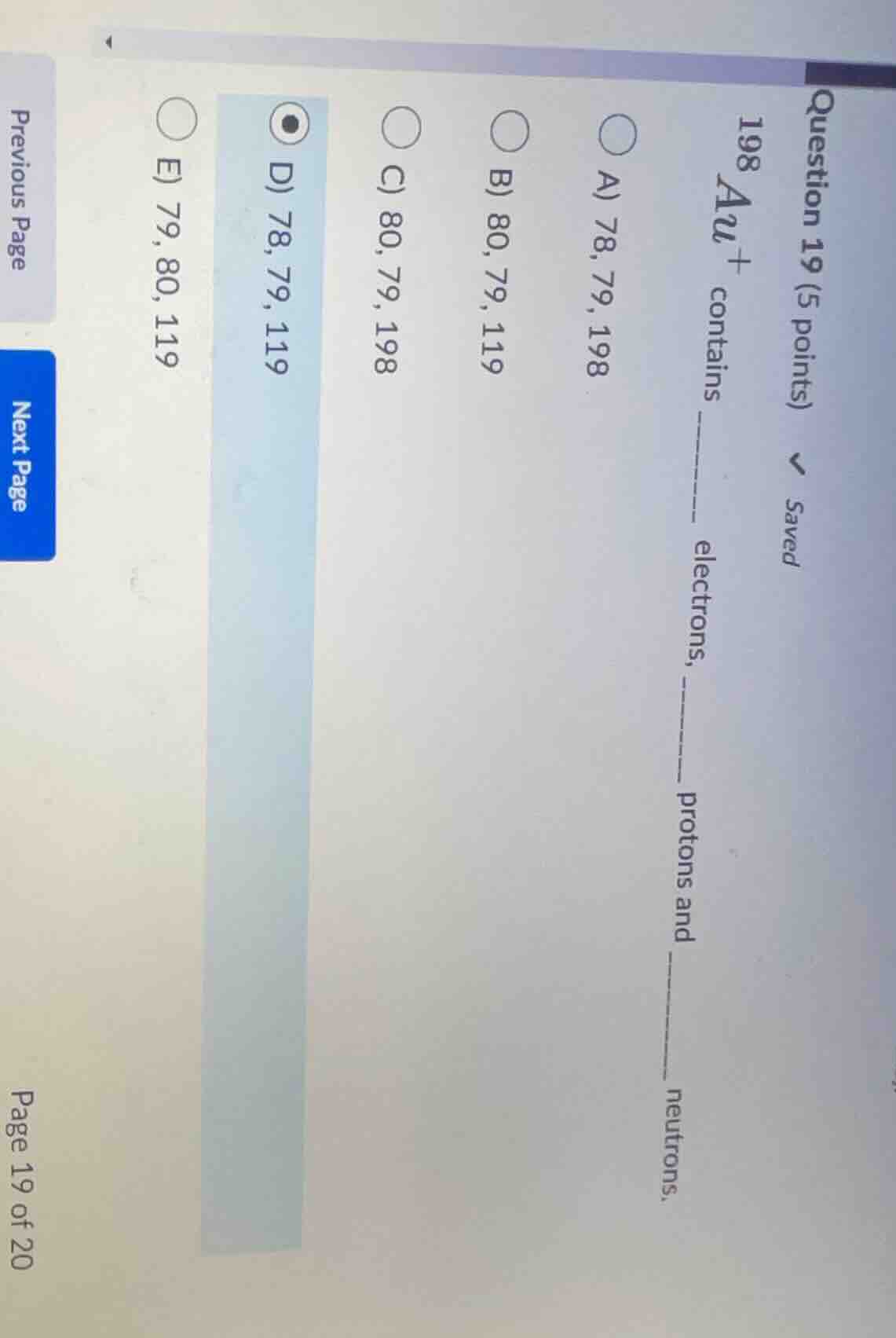
question 19 (5 points) \\(^{198}_{}au^+\\) contains ____ electrons, __ protons and ____ neutrons. a) 78, 79, 198 b) 80, 79, 119 c) 80, 79, 198 d) 78, 79, 119 e) 79, 80, 119
Step1: Determine atomic number of Au
Gold (Au) has an atomic number (number of protons) of 79. So protons = 79.
Step2: Find electrons in \( \ce{^{198}Au+} \)
For a cation \( \ce{Au+} \), it has lost 1 electron. Neutral Au has electrons = protons = 79, so \( \ce{Au+} \) has \( 79 - 1 = 78 \) electrons.
Step3: Calculate neutrons
Mass number (198) = protons + neutrons. So neutrons = \( 198 - 79 = 119 \).
So electrons = 78, protons = 79, neutrons = 119, which matches option D.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D) 78, 79, 119