QUESTION IMAGE
Question
question 31 of 60
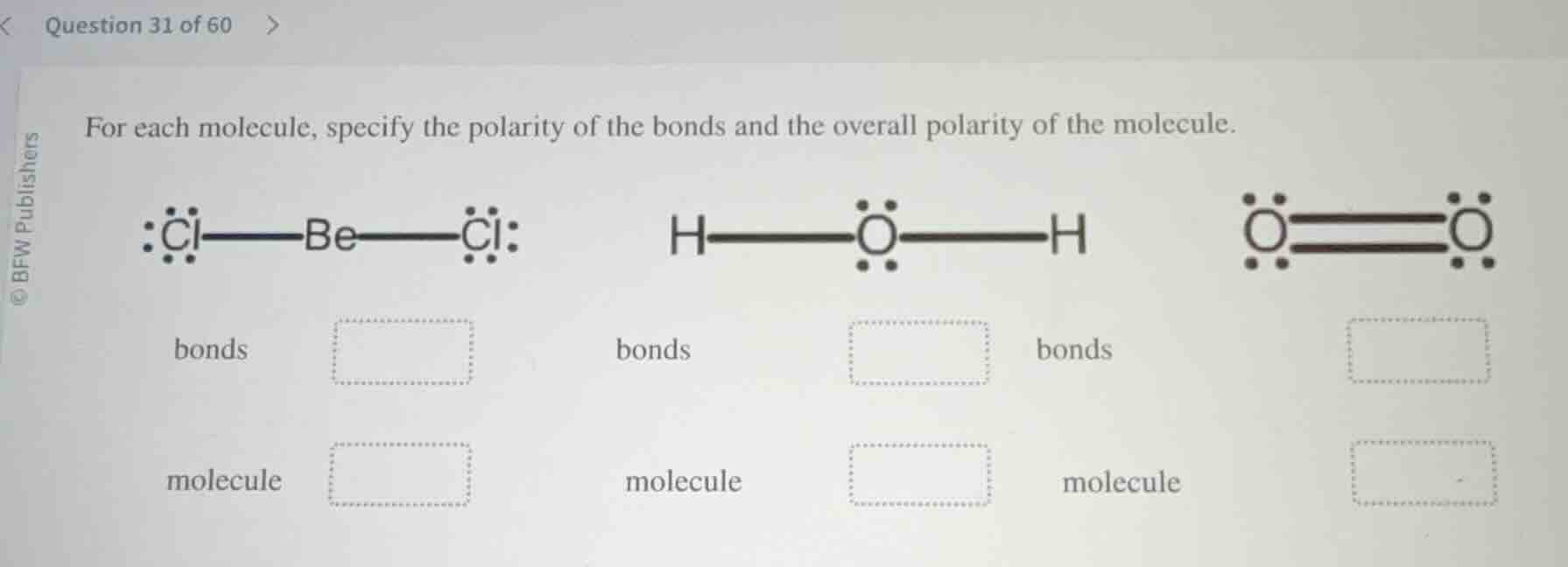
for each molecule, specify the polarity of the bonds and the overall polarity of the molecule.
bonds
molecule
bonds
molecule
bonds
molecule
Brief Explanations
- For $\text{BeCl}_2$:
- Bond polarity: Beryllium (Be) and chlorine (Cl) have different electronegativities, so $\text{Be-Cl}$ bonds are polar.
- Molecular polarity: The molecule has a linear, symmetrical geometry. The polar bond dipoles point in opposite directions and cancel each other out, making the overall molecule nonpolar.
- For $\text{H}_2\text{O}$:
- Bond polarity: Hydrogen (H) and oxygen (O) have different electronegativities, so $\text{O-H}$ bonds are polar.
- Molecular polarity: The molecule has a bent, asymmetrical geometry. The polar bond dipoles do not cancel, creating a net molecular dipole, so the overall molecule is polar.
- For $\text{O}_2$:
- Bond polarity: The bond is between two identical oxygen atoms, which have identical electronegativities, so the $\text{O=O}$ bond is nonpolar.
- Molecular polarity: The molecule is symmetrical, and with nonpolar bonds, there is no net dipole, so the overall molecule is nonpolar.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{BeCl}_2$
- bonds: Polar
- molecule: Nonpolar
- $\text{H}_2\text{O}$
- bonds: Polar
- molecule: Polar
- $\text{O}_2$
- bonds: Nonpolar
- molecule: Nonpolar