QUESTION IMAGE
Question
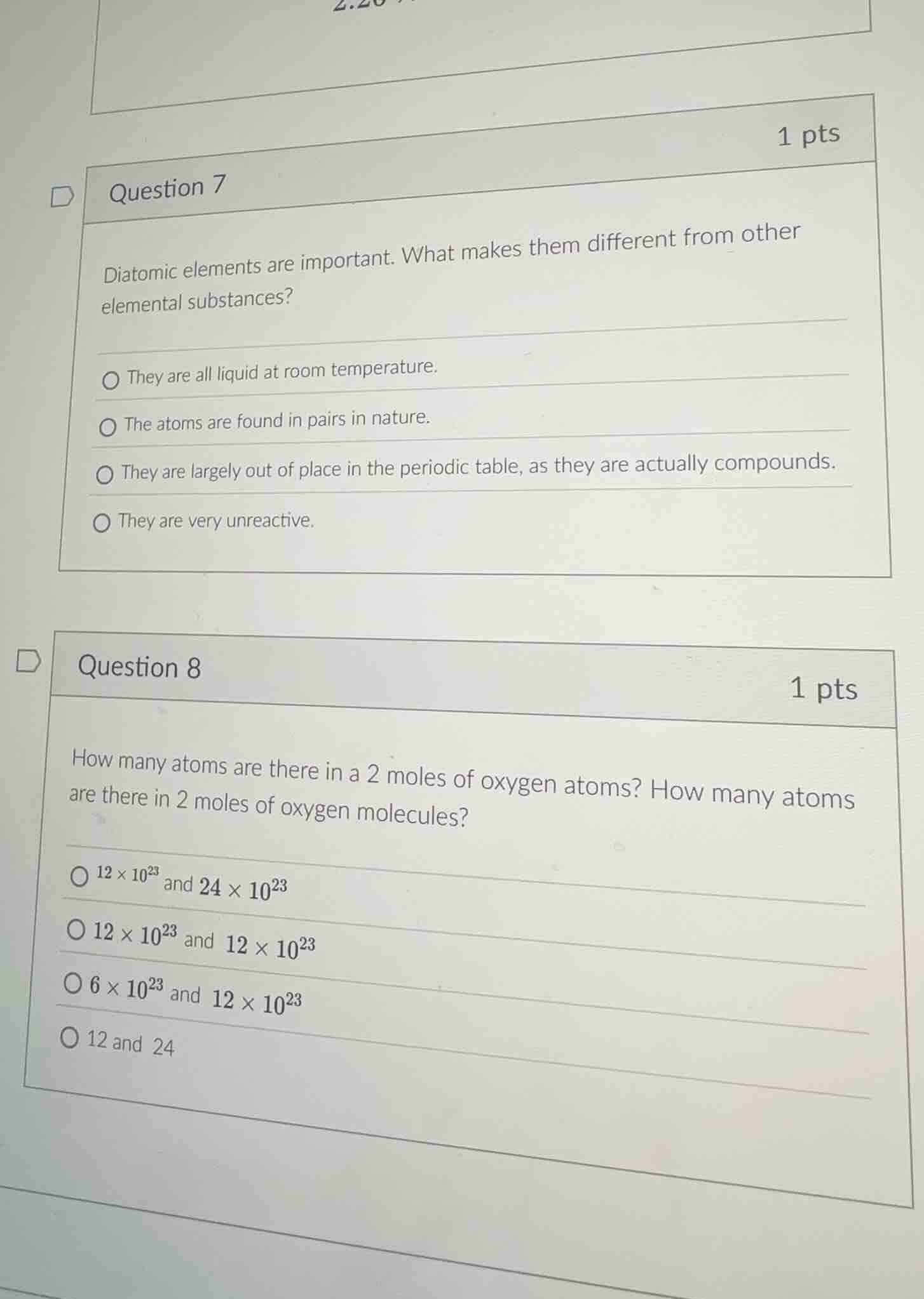
question 7
diatomic elements are important. what makes them different from other elemental substances?
they are all liquid at room temperature.
the atoms are found in pairs in nature.
they are largely out of place in the periodic table, as they are actually compounds.
they are very unreactive.
question 8
how many atoms are there in a 2 moles of oxygen atoms? how many atoms are there in 2 moles of oxygen molecules?
$12 \times 10^{23}$ and $24 \times 10^{23}$
$12 \times 10^{23}$ and $12 \times 10^{23}$
$6 \times 10^{23}$ and $12 \times 10^{23}$
12 and 24
Question 7
- Option 1: Diatomic elements like hydrogen, oxygen are gaseous at room temp, so wrong.
- Option 2: Diatomic elements exist as diatomic molecules (atoms in pairs, e.g., \(O_2\), \(H_2\)), this is correct.
- Option 3: Diatomic elements are elements, not compounds, so wrong.
- Option 4: Many diatomic elements (like oxygen, hydrogen) are reactive, so wrong.
Step1: Recall Avogadro's number (\(N_A = 6\times10^{23}\) atoms/mol or molecules/mol)
Step2: For 2 moles of oxygen atoms:
Number of atoms = moles \(\times N_A\) = \(2 \times 6\times10^{23} = 12\times10^{23}\)
Step3: For 2 moles of oxygen molecules (\(O_2\)):
Each \(O_2\) has 2 O atoms. Number of molecules = \(2 \times 6\times10^{23}\), so number of atoms = \(2 \times 2 \times 6\times10^{23} = 24\times10^{23}\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The atoms are found in pairs in nature.