QUESTION IMAGE
Question
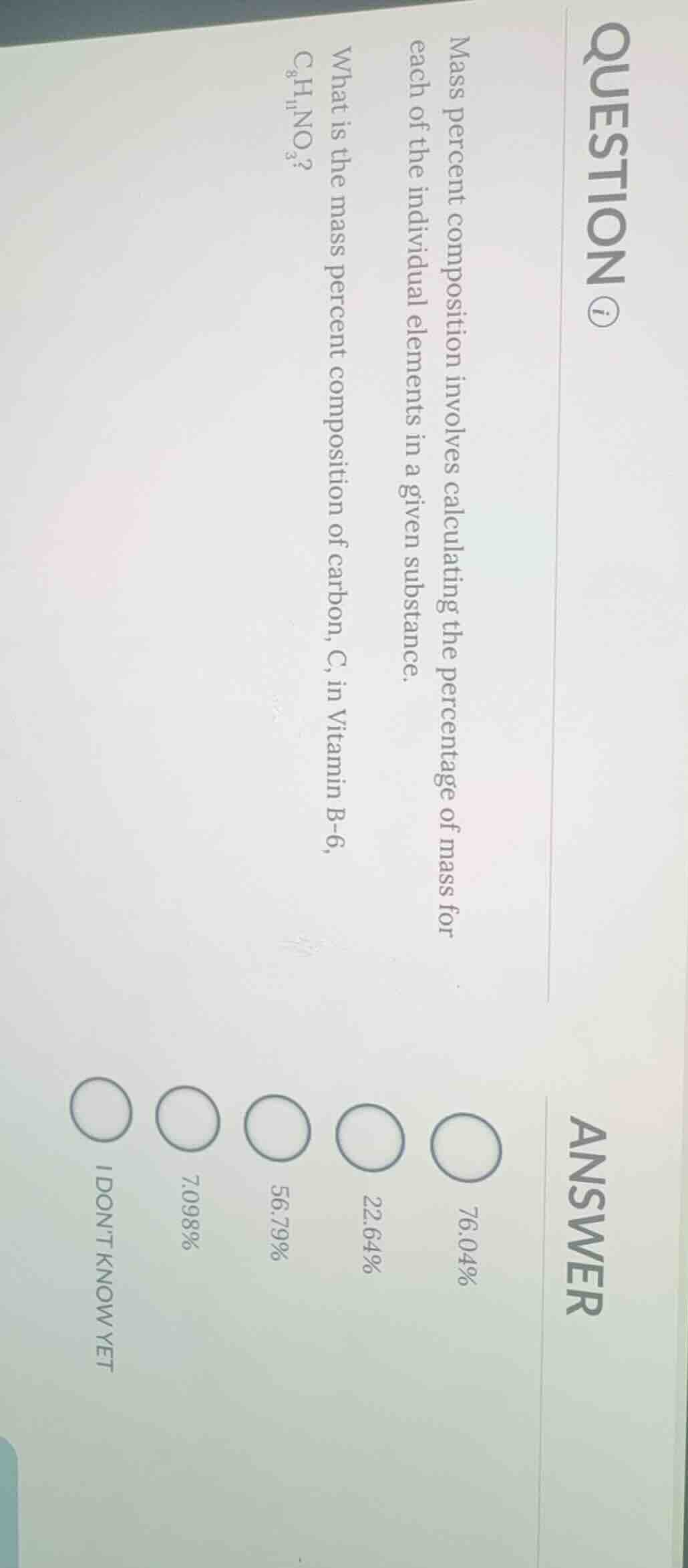
question ⓘ
mass percent composition involves calculating the percentage of mass for each of the individual elements in a given substance.
what is the mass percent composition of carbon, c, in vitamin b-6, $ce{c_{8}h_{11}no_{3}}$?
answer
○ 76.04%
○ 22.64%
○ 56.79%
○ 7.098%
○ i dont know yet
Step1: Calculate molar mass of C
Molar mass of C = $12.01\ \text{g/mol}$, 8 C atoms:
$8 \times 12.01 = 96.08\ \text{g/mol}$
Step2: Calculate total molar mass of $\text{C}_8\text{H}_{11}\text{NO}_3$
Molar masses: H=$1.008$, N=$14.01$, O=$16.00$
$$8(12.01) + 11(1.008) + 14.01 + 3(16.00)$$
$$= 96.08 + 11.088 + 14.01 + 48.00 = 169.178\ \text{g/mol}$$
Step3: Calculate mass percent of C
Mass percent = $\frac{\text{Total mass of C}}{\text{Molar mass of compound}} \times 100\%$
$$\frac{96.08}{169.178} \times 100\% \approx 56.79\%$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. 56.79%